Does LAH react with esters
* LiAlH4 reagent can reduce aldehydes to primary alcohols, ketones to secondary alcohols, carboxylic acids and esters to primary alcohols, amides and nitriles to amines, epoxides to alcohols and lactones to diols. * Lithium aluminium hydride, LAH reagent cannot reduce an isolated non-polar multiple bond like C=C.
Can LiAlH4 reduce ester?
LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols. Amides and nitriles are reduced to amines.
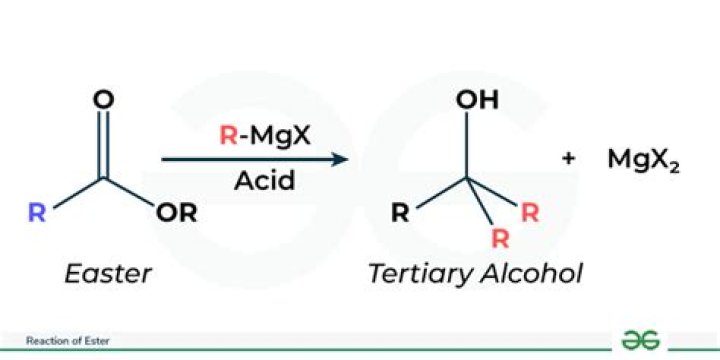
When an ester is reacted with LAH which functional group will be formed?
Esters can undergo hydride reduction with LiAlH4 to form two alcohols. The alcohol derived from the acyl group of the ester will be 1o and is typically considered the main product of the reaction. The other alcohol is derived from the ester’s alkoxy group and is typically considered a side-product of the reaction.
What does LAH do as a reagent?
Lithium aluminum hydride (LiAlH4) is a strong reducing agent. It will reduce almost any C=O containing functional group to an alcohol. One equivalent of H- adds, and then another equivalent adds, unavoidably.Can LAH reduce ethers?
Lithium Aluminium Hydride (LiAlH4, LAH) – much more reactive than NaBH4. … LiAlH4 (in ether) reduces aldehydes, carboxylic acids, and esters to 1° alcohols and ketones to 2° alcohols.
Does lah reduce carboxylic acids?
Thanks to its high reactivity, LAH easily reduces all classes of carboxylic acid derivatives, generally to the –1 oxidation state. Acids, esters, anhydrides and acyl chlorides are all reduced to 1º-alcohols, and this method is superior to catalytic reduction in most cases.
Does LAH reduce alkynes?
Tertiary halides react only in certain cases. Lithium aluminium hydride does not reduce simple alkenes or arenes. Alkynes are reduced only if an alcohol group is nearby.
Why is lah stronger than NaBH4?
Lithium aluminium hydride (LiAlH4) is more electropositive (more metallic) than boron in NaBH4. The hydride from LiAlH4 is therefore more electron rich and thus is a stronger base (in reaction with water) and stronger nucleophile (with carbonyl group).What can reduce esters?
Esters are normally reduced by reaction with lithium aluminum hydride.
Can b2h6 reduce Ester?Diborane has been known to be an effective reagent for reductions of aldehydes and ketones to borate esters, which give alcohols on quenching. In the case of aryl carbonyl functions, reduction with this electrophilic reagent is closely related to those mentioned in Section 1.13.
Article first time published onIs lah a LiAlH4?
Lithium aluminum hydride (LiAlH4; LAH): A hydride source used primarily for reduction of carbonyl compounds. … Lithium aluminum hydride reduces a ketone to a secondary alcohol. Lithium aluminum hydride reduces an ester to two alcohols. Lithium aluminum hydride reduces a tertiary amide to a tertiary amine.
How do you quench the LAH reaction?
To quench your LAH reduction, add the celite/ hydrated sodium sulfate mixture to your reaction mixture one spatula full at a time. This will be exothermic and might cause the reaction to temporarily stop stirring, but keep adding the solid until the mixture resumes stirring.
Does lah reduce double bonds?
Lithium aluminium hydride (LiAlH4) is a strong reducing agent. It cannot reduce an isolated non-polar multiple bond like C=C. However, the double bonds are reduced when a phenyl group is attached to the beta-carbon atom. Lithium aluminium hydride (LiAlH4) is a strong reducing agent.
What happens when ester reacts with a base?
Basic hydrolysis of an ester gives a carboxylate salt and an alcohol.
What type of reaction is esterification of amides reversible or irreversible?
Explanation: Esterification of Amides is a reversible reaction.
Can H2 PD reduce ketones?
We now demonstrate that aryl aldehydes and ketones as well as aryl epoxides can also effectively be reduced using Pd(0) EnCat™ 30NP under conventional catalytic hydrogenation conditions of H2 (atmospheric pressure) with good selectivity and conversions [20].
What can H2 NI reduce?
H2 Ni are strong reducing agents which reduces aldehydes to primary alcohols,ketones to secondary alcohols and carboxylic acid and its derivatives to primary alcohols. H2 and Ni usually are used for the hydrogenation of Alkenes and alkynes.
What does H3O+ do to an alkene?
Ch 6: Alkene + H3O+ When treated with aq. acid, most commonly H2SO4, alkenes form alcohols. Reaction proceeds via protonation to give the more stable carbocation intermediate.
Why does lah react violently with water?
Solvents for Lithium Aluminum Hydride and Sodium Borohydride Both aluminum hydride and borohydride react with protic solvents such as water and ethanol. Lithium aluminum hydride reacts violently with water to form hydrogen gas, which may burn explosively because of the heat generated in the reaction.
Why NaBH4 Cannot reduce Ester?
At this point, the reactivity is too weak to occur without a stronger reducing agent. The short answer is that NaBH4 is simply too weak a reducing agent for carboxylic acids and esters and the two will simply mingle in solution with little to no reaction for months.
Does NaBH4 reduce alkynes?
Alkenes and alkynes are rapidly reduced to the corresponding alkanes using sodium borohydride and acetic acid in the presence of a small amount of palladium catalyst. The heterogeneous reaction is conducted in open air at room temperature.
Is lah a metal hydride?
Therefore, the use of metal hydrides are the preferred alternative reducing agents. Two valuable reducing agents commonly used are lithium aluminum hydride (LAH) and sodium borohydride (NBH). Both reduce aldehydes and ketones to alcohols. These two metal hydrides are quite different in their reactivities.
Which reagent will bring about the conversion of carboxylic acid into ester?
Conversion to acid derivatives Esters can be prepared by treatment of a carboxylic acid with an alcohol in the presence of an acid catalyst, most commonly sulfuric acid or hydrochloric acid, in a reaction known as Fischer esterification.
What reagent reduces carboxylic acids?
Although carboxylic acids are more difficult to reduce than aldehydes and ketones, there are several agents that accomplish this reduction, the most important being lithium aluminum hydride (LiAlH4) and borane (BH3). The product is a primary alcohol (RCOOH → RCH2OH).
What happens when reduction of carboxylic acid with lia1h4?
Carboxylic acids can be converted to 1o alcohols using Lithium aluminum hydride (LiAlH4). … An aldehyde is produced as an intermediate during this reaction, but it cannot be isolated because it is more reactive than the original carboxylic acid.
Does H2 PD reduce esters?
Reduce aldehyde or ketone to alcohol: use H2 with Raney nickel iv. Reduce acid chloride to aldehyde: use a partially deactivated Pd catalyst (this is the Rosenmund reduction) v. … It will not reduce an acid or an ester.
Can Dibal reduce esters?
DIBAL can be used to reduce many a functional group, but it is most commonly used to reduce carboxylic acid esters to aldehydes, which can not be done using lithium aluminumhydride, the traditional reducing agent used to reduce carbonyl compounds.
Why is libh4 a stronger reducing agent than NaBH4?
But LiAlH4 is a very strong reducing agent than NaBH4 because the Al-H bond in the LiAlH4 is weaker than the B-H bond in NaBH4. … Therefore, the low electronegativity shifts the electron density towards the hydrogen in Al-H than that of B-H bond. As a result, LiAlH4 is a better hydride donor.
Why is LiAlH4 used in dry ether?
The hydride ion in LiAlH4 is very basic. For this reason, LiAlH4 reacts violently with water and therefore must be used in dry solvents such as anhydrous ether and THF. … The lithium ion acts as a Lewis acid catalyst by coordinating to the carbonyl oxygen.
Which one of the following reagents would convert esters into aldehydes?
Esters can be converted to aldehydes using diisobutylaluminum hydride (DIBAH). The reaction is usually carried out at -78 oC to prevent reaction with the aldehyde product.
What does B2H6 reduce?
Borane is commonly used for the reduction of carboxylic acids in the presence of esters, lactones, amides, halides and other functional groups. In addition, borane rapidly reduces aldehydes, ketones, and alkenes. … In addition, though highly flammable, gaseous diborane (B2H6) is available.



