How do electrons transfer
Whenever electrons are transferred between objects, neutral matter becomes charged. For example, when atoms lose or gain electrons they become charged particles called ions. Three ways electrons can be transferred are conduction, friction, and polarization. In each case, the total charge remains the same.
How are electrons transferred in a covalent bond?
A covalent bond consists of the mutual sharing of one or more pairs of electrons between two atoms. These electrons are simultaneously attracted by the two atomic nuclei. A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions.
What electrons are transferred?
The attraction between oppositely charged ions is called an ionic bond, and it is one of the main types of chemical bonds in chemistry. Ionic bonds are caused by electrons transferring from one atom to another.
Where does electron transfer occur?
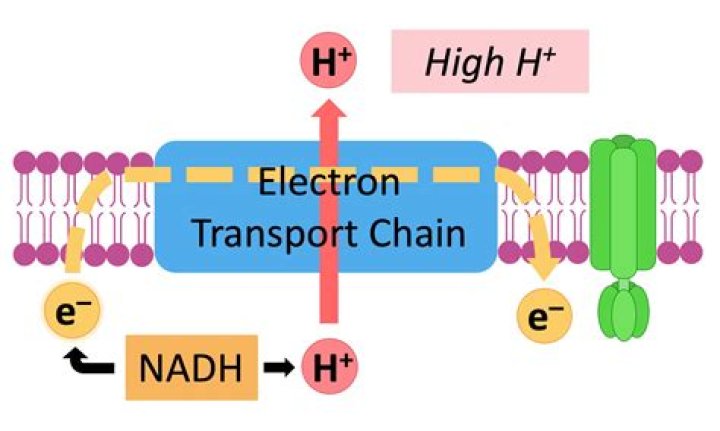
Electron transport chain ETC consists in electron transport via reduction and oxidation (redox) reactions happening simultaneously through a series of carrier proteins inserted into the inner mitochondrial membrane.What kind of element gains electron after ionic bonding?
An atom that loses one or more valence electrons to become a positively charged ion is known as a cation, while an atom that gains electrons and becomes negatively charged is known as an anion.
Which compound is formed by transferring electron?
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion.
Why are atoms attracted in an ionic bond?
Ionic bonds involve a cation and an anion. The bond is formed when an atom, typically a metal, loses an electron or electrons, and becomes a positive ion, or cation. … The ions produced are oppositely charged and are attracted to one another due to electrostatic forces.
Which atoms will bond when valence electrons are transferred from one atom to the other?
Ionic bonding Definition: An ionic bond is formed when valence electrons are transferred from one atom to the other to complete the outer electron shell. Example: A typical ionically bonded material is NaCl (Salt): The sodium (Na) atom gives up its valence electron to complete the outer shell of the chlorine (Cl) atom.When an atom gains an electron what does it become?
If a neutral atom gains an electron, it becomes negative. This kind of ion is called an anion. If a neutral atom loses an electron, it becomes positive. This kind of ion is called a cation.
What happens when an atom gains an electron?If an atom or molecule gains an electron, it becomes negatively charged (an anion), and if it loses an electron, it becomes positively charged (a cation). Energy may be lost or gained in the formation of an ion.
Article first time published onHow do electrons behave differently in ionic and covalent bonds?
In ionic bonding, atoms transfer electrons to each other. Ionic bonds require at least one electron donor and one electron acceptor. In contrast, atoms with the same electronegativity share electrons in covalent bonds, because neither atom preferentially attracts or repels the shared electrons.
What type of electrons are involved in bonding?
The electrons on the outermost energy level of the atom are called valence electrons. The valence electrons are involved in bonding one atom to another. The attraction of each atom’s nucleus for the valence electrons of the other atom pulls the atoms together.
What types of atoms are held together in an ionic bond?
An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. A covalent bond involves a pair of electrons being shared between atoms.
Does transferring an electron change the identity of an element?
Does transferring an electron change the identity of the elements involved? Explain. Changing the number of electrons does not change the identity of the atom; only the charge changes. In order to change the identity of an atom, you need to change the number of protons in the nucleus.
When an atom loses an electron what becomes a cation?
When an atom loses an electron The atom becomes? If an atom has an equal number of protons and electrons, its net charge is 0. If it gains an extra electron, it becomes negatively charged and is known as an anion. If it loses an electron, it becomes positively charged and is known as a cation.
Which atoms will bond when valence electrons are transferred from one atom to the other 1 2 3 O and Se O and Sr O and HO and P?
QuestionsAnswerLinks12 Which atoms will bond when valence electrons are transferred from one atom to the other? (1) O and Se (3) O and H (2) O and Sr (4) O and P2link13 Which sample of matter is a mixture? (1) Br2(ℓ) (3) KBr(s) (2) K(s) (4) KBr(aq)4link
What kind is it when one atom takes an electron from another atom?
In ionic bonding, electrons are completely transferred from one atom to another. In the process of either losing or gaining negatively charged electrons, the reacting atoms form ions. The oppositely charged ions are attracted to each other by electrostatic forces, which are the basis of the ionic bond.
Why does an atom lose energy when it gains an electron?
An atom gains electron to attain stable electronic configuration. As stability is related to energy , more stable less energy. Hence when an atom gains electron it loses energy to attain stability.
What makes ionic bonds different from covalent bonds?
Differences between ionic and covalent bonds In covalent bonds, atoms share electrons, whereas in ionic bonds atoms transfer electrons. The reaction components of covalent bonds are electrically neutral, whereas for ionic bonds they are both charged.
Are ionic bonds conductive?
Ionic compounds conduct electricity when molten (liquid) or in aqueous solution (dissolved in water), because their ions are free to move from place to place. … Ionic compounds are conductors of electricity when molten or in solution, and insulators when solid.
How are covalent and ionic bonds the same?
An ionic bond essentially donates an electron to the other atom participating in the bond, while electrons in a covalent bond are shared equally between the atoms. The only pure covalent bonds occur between identical atoms. … Ionic bonds form between a metal and a nonmetal. Covalent bonds form between two nonmetals.
Which electrons are available for bonding and why?
Valence electrons are the electrons in the outermost shell of an atom and they are the electrons available for forming chemical bonds with other atoms.
How does ionic bonding hold particles together?
An ionic bond is held together by the electrostatic attraction between ions that are near one another. Electrostatic attraction is the attraction between atoms that have opposite charge and holds the atoms together in ionic bonds. … The atom actually “accepts” or “takes” the electrons that the other atom is giving up.



