How does soda ash raise pH
When you add Soda Ash to the pool, the carbonate anion (CO32-) wants to become bicarbonate (HCO3-). To do so, it needs to get a Hydrogen (H+) from somewhere. When carbonate takes its Hydrogen atom, the pH goes up.
How does soda ash affect pH?
Soda ash raises pH and alkalinity. Usually, if you try to use it to raise pH, you will raise the alkalinity too high and have to add acid to adjust it back down.
How does soda ash increase alkalinity?
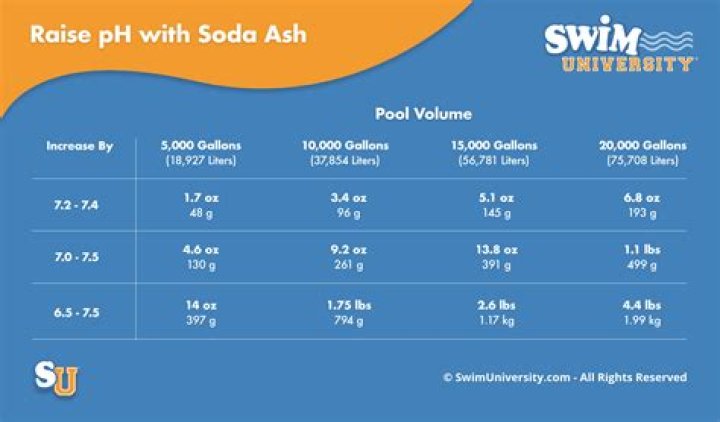
Fill a container with soda ash and walk around the circumference of the pool, distributing the soda ash into the middle of the pool. Add 1.6 ounces of soda ash for every 1,000 gallons of water in the pool and for every 10 parts per million of increase in the total alkalinity that is needed.
Does soda ash raise pH and alkalinity?
The industry standard has always been to use sodium bicarbonate (baking soda) to raise total alkalinity and sodium carbonate (soda ash) to raise pH — the exception being if both total alkalinity and pH are low. … Sodium carbonate will actually have a dramatic effect on both pH and total alkalinity.How much soda ash do I add to raise pH?
To raise the pH of your pool, fill a 5 gallon bucket with water. Then, add 6 ounces of soda ash to the bucket for every 10,000 gallons of water in the pool. Pour that mixture around the pool’s perimeter while the filter is running so it mixes evenly. After an hour, check the pools pH.
Will soda ash raise pH in pool?
To raise your pool pH levels, try adding in sodium carbonate (AKA soda ash). Make sure that you don’t add any more than two pounds of soda ash per 10,000 gallons of water per treatment. … Expect your pool water to cloud up after you’ve added in the soda ask. However, your water will turn clear after a day or two!
Does sodium bicarbonate raise pH?
Baking soda, also known as sodium bicarbonate is naturally alkaline, with a pH of 8. When you add baking soda to your pool water, you will raise both the pH and the alkalinity, improving stability and clarity.
Is soda ash the same thing as baking soda?
Baking soda, known as sodium bicarbonate (NaHCO3) is composed of one atom of sodium, one atom of hydrogen, one atom of carbon and three atoms of oxygen. Soda ash, known as sodium carbonate (Na2CO3) is made from two atoms of sodium, one atom of carbon and three atoms of oxygen.Can I swim after adding soda ash?
It is recommended to wait at least 20 minutes to an hour after adding water balancing chemicals. You should wait 2-4 hours (or one full cycle through the filter) to swim from the moment you use calcium chloride in your pool.
How do you mix soda ash with water?Mix 2 lbs of Soda Ash with 5 gallons of water. Set your pump at 50% or 60% capacity. Let the unit run for a time, and use water as you normally would, then check the pH a few feet downstream from the injection point. If the pH isn’t where you want it, make adjustments.
Article first time published onHow do you raise pH but not alkalinity?
When both Total Alkalinity and pH are too low (TA less than 80 ppm, pH below 6.8), use pH Increaser (sodium carbonate) to bring both levels up. If your Total Alkalinity levels are too high, add pH Reducer (sodium bisulfate).
What raises pH in pool?
The two primary chemicals to increase pH are sodium carbonate, aka soda ash (Na2CO3), and sodium bicarbonate (NaHCO3). Both raise the alkalinity, but soda ash has a stronger impact on raising the pH.
How much baking soda does it take to raise pH?
As per standard, 1.25 pounds of baking soda is enough to raise the PH level of a 10,000-gallon pool by 10ppm, so to achieve a 100ppm alkalinity, you would need 12.5 pounds of baking soda for a 100,000 gallon of pool water.
How much does NaOH increase pH?
In the above example 10 ml or NaOH was added to increase the pH from 2.0 to 3.0. An additional 1 ml was added to further raise the pH to 4.0, and another 0.1 ml added to raise the pH to 5.0.
Does muriatic acid lower pH?
When used correctly, muriatic acid can lower the pH levels of your pool water. The potency of muriatic acid makes it beneficial at getting rid of hardened water that has become too alkaline.
How long does it take for soda ash to clear?
#5 Cause: Adding Soda Ash or Baking Soda to the pool. When you add a base to the pool it can become cloudy for a short period of time. After a few hours or by the next day it should clear up.
Is sodium bicarbonate acidic or alkaline?
Sodium bicarbonate is a salt that breaks down to form sodium and bicarbonate in water. This breakdown makes a solution alkaline, meaning it is able to neutralize acid. Because of this, sodium bicarbonate is often used to treat conditions caused by high acidity in the body, such as heartburn.
What is pH of sodium bicarbonate?
Sodium bicarbonate is a white powder with molecular formula NaHCO3, and pH of 8.5 (1% aqueous solution, 25°C). Among sodium compounds, it is one of the substances with the lowest alkalinity.
Does shock raise pH?
When you shock a pool, you test and adjust the pH level for a reason. With that said, if you shock a pool outside of the 7.2 to 7.4 pH range, not only will you waste a significant amount of the chlorine used, you will also end up with cloudy water.
Does sodium carbonate raise alkalinity?
Sodium carbonate will actually have a dramatic effect on both pH and total alkalinity. … On the other hand, 12.2 ounces of sodium carbonate will raise the pH of 10,000 gallons of water by 0.4, as well as increase the total alkalinity by roughly 8.6 ppm.
Does soda ash go bad?
Yes, you can save soda ash solutions indefinitely. Soda ash never goes bad. Mold will not grow in it, and it does not degrade chemically to something else. You can reuse it for presoaking your clothes for tie-dyeing until there is no more left in the bucket.
Will chlorine raise pH?
Using liquid chlorine raises the pH of the water. Liquid chlorine does not raise pH. When added to water, liquid chlorine (which has a pH of 13) makes HOCl (hypochlorous acid – the killing form of chlorine) and NaOH (sodium hydroxide), which raises pH. … So the net effect on pH is zero (or almost zero).
Why do you add soda ash to your pool?
Soda ash can do just that. Also known as sodium carbonate, it raises both the pH level and total alkalinity of the pool when added to the water. Note: The ideal pH level for a pool is between 7.4 and 7.6. Anything over this number and the water is too alkaline (or basic), while anything under is too acidic.
How do I lower my pH?
To bring down pH, use a made-for-pools chemical additive called pH reducer (or pH minus). The main active ingredients in pH reducers are either muriatic acid or sodium bisulfate (also called dry acid). Reducers are readily available at pool supply stores, home improvement centers and online.
Can you eat soda ash?
Ingestion of the concentrated salt or of strong cleaning solutions can cause nausea, vomiting, stomach ache, diarrhea and burns to the mouth and throat.
What does pH increaser do?
pH & Alkalinity Increaser is sodium bicarbonate (also called sodium hydrogen carbonate). It raises Total Alkalinity, and pH which is too low. Since it has a pH of only 8.3, it will generally have a lesser effect on pH.
What is the difference between soda ash and alkalinity up?
Soda ash will raise pH and minimally raise alkalinity, while baking soda will raise alkalinity and minimally raise pH. Use baking soda when the pH isn’t off by much, but the alkalinity is way too low, and use soda ash when the pH needs to be drastically increased but you want the alkalinity to be less affected.
Is soda ash a water softener?
Lime Soda Ash Softening. Chemical precipitation is one of the more common methods used to soften water. Chemicals normally used are lime (calcium hydroxide, Ca(OH)2) and soda ash (sodium carbonate, Na2CO3).
What happens if you don't use soda ash when tie dying?
We use it to elevate the pH so that the dye can react with the fibers. It’s only in the presence of an alkaline environment that the dyes are able to react at room temperature. Forgetting to use soda ash will leave you with very pale colors.
What does Borax do in a pool?
Borax acts as an effective pH buffer and helps prevent algae growth in swimming pools. Plus, it will leave your water looking sparkly and feeling soft.
What is more important pH or alkalinity?
Regardless of the starting pH, the higher the alkalinity of the water source, the more acid is required to reduce pH to 5.0. For all water sources, it is the alkalinity that actually determines how much acid to use, not the pH.