How is chlorine gas stored
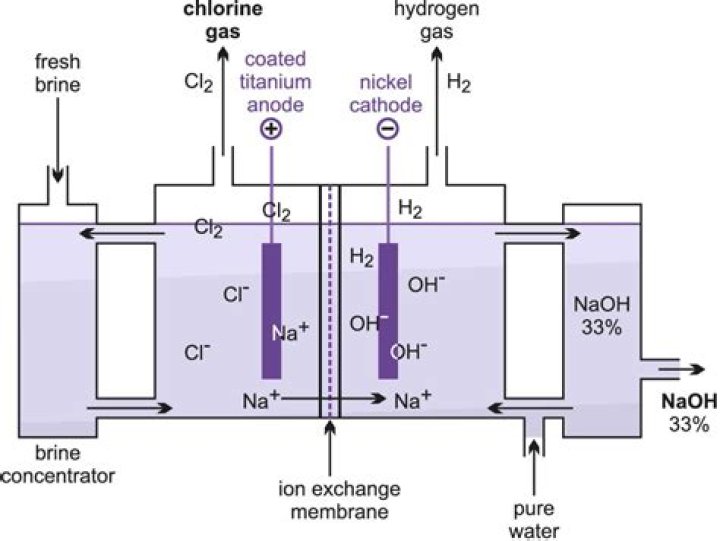
Chlorine gas, also called elemental chlorine, is stored in cylinders as a liquefied compressed gas. It is toxic and irritating to skin, eyes, nose, and mucous membranes. In strong concentrations it is visible as a greenish-yellow cloud.
How do you manage chlorine gas?
Quickly moving to an area where fresh air is available is highly effective in reducing exposure to chlorine. If the chlorine release was outdoors, move away from the area where the chlorine was released. Go to the highest ground possible, because chlorine is heavier than air and will sink to low-lying areas.
Does chlorine gas degrade?
When sodium hypochlorite is used, acetic or sulfuric acid is added to the water. An excessive dose can produce poisonous gases. … After those reactions are complete, chlorine will slowly escape into the air as a gas. For this reason, free and total chlorine levels slowly degrade over time in a container.
How long does chlorine gas stay in the air?
It depends on the wind and weather. ANY release of chlorine gas should be reported to emergency services. When treated effluent is released into receiving waters, free residual chlorine dissipates rapidly (it has a half-life of 1.3 to 5 hours).How is liquid chlorine stored?
The safest option for storing pool chemicals is to put the sealed containers into hard plastic bins with tight-fitting lids. Ideally, use several bins so that you can separate liquids and solids, and so that reactants such as chlorine and muriatic acid can be stored separately.
How is chlorine gas made?
The combination of bleach (sodium hypochlorite) with acid produces chlorine gas, a heavy green-yellow gas with a strong odor. … Chlorine gas is pressurized and cooled for easy storage in liquid form. When released, the liquid form of chlorine quickly turns into yellow-green colored gas with an irritating odor.
What Temperature Does chlorine turn into a gas?
PropertyDefinitionConditionsGas32°F, 14.696 psia (0°C, 101.325 kPa)Saturated Gas32°F (0°C)Saturated Liquid32°F (0°C)Vapor PressureThe absolute pressure of chlorine gas above liquid chlorine when they are in equilibrium32°F (0°C) 77°F (25°C)
How do you remove chlorine gas from the air?
Emergency gas scrubbers are incredibly effective at removing chlorine gas from the air while ensuring the safety of personnel and surrounding communities. Dry scrubbers utilize gas phase filtration to rid the air of toxic gases in the event of a release.What neutralizes chlorine gas?
Chlorine in a swimming pool is most commonly reduced or neutralized by adding sodium thiosulfate (Na2S2O3) to the pool water.
How much chlorine gas is lethal?Concentrations of about 400 ppm and beyond are generally fatal over 30 minutes, and at 1,000 ppm and above, fatality ensues within only a few minutes.
Article first time published onDoes chlorine gas dissolve in water?
Chlorine is slightly water soluble, and reacts with moisture to form hypochlorous acid (HClO) and hydrochloric acid (HCl).
How do you turn liquid chlorine into gas?
Generate a gentle stream of chlorine by dripping the hydrochloric acid slowly on to the sodium chlorate(I). The greenish-yellow gas will gradually fill both flasks. After about a minute, yellow drops of liquid chlorine begin to condense on the ‘cold finger’ and drop onto the bottom of the flask.
Is chlorine and chlorine gas the same?
AluminiumSiliconPhosphorusChlorine
How is liquid chlorine transported?
Chlorine is typically shipped and stored as a liquid in a container under pressure. … Tank cars shipped by motor vehicle may have a capacity up to 22 tons. Under ambient temperature and not under pressure, chlorine is a gas.
Can you store chlorine in the garage?
Pool chemicals should be stored in a cool, dry, well-ventilated place. Garages are generally not the best place to store pool chemicals unless they are locked in a storage bin or cabinet. Oxidizers and strong acids corrode metal and can cause heavy rusting of pool, electrical, and other equipment stored in the room.
Why is there a storage of chlorine?
The shortage is due to increased demand for pool supplies during the pandemic and a chemical fire at a BioLab facility in Louisiana after Hurricane Laura that knocked out one of the country’s three main chlorine manufacturers.
Why is chlorine gas so reactive?
Halogens are notorious electron-hogs; powerfully attracting electrons from atoms of other elements, particularly from the alkali metals. This makes the halogens highly reactive. Chlorine, being one of the smaller halogens, will react strongly with most elements.
Why is chlorine a gas at room temp?
In Cl2 there are covalent bonds between the atoms forming simple molecules. There are weak attractions between molecules of Cl2 meaning little energy is needed to break these forces of attraction and therefore Cl2 has a low boiling point.
Why chlorine is written as Cl2?
Chlorine is called Cl2 because it is a diatomic molecule. Diatomic means there are two atoms of the same elements in the molecule. There are other diatomic molecule, such as hydrogen (H2), nitrogen (N2), oxygen (O2), fluorine (F2), chlorine (Cl2), bromine (Br2), iodine (I2).
How do you turn chlorine into salt?
Salt systems are actually chlorine generators, using a process called electrolysis. Electrolysis happens by sending electricity through salt water (sodium chloride, or NaCl), which interacts with the chloride ion in the salt. This creates chlorine in the water, and a very high-pH byproduct called sodium Hydroxide.
What happens when you mix bleach and ammonia?
When bleach is mixed with ammonia, toxic gases called chloramines are produced. Exposure to chloramine gases can cause the following symptoms: Coughing. Nausea.
Is bleach flammable with heat?
While it is used all over, many don’t know what bleach is made from and they wonder if it can catch on fire or even explode if heated. Household bleach (like Clorox) is not considered a flammable or explosive substance by itself.
How do you get rid of chlorine without oxygen?
Chlorine can also be used to disinfect water by pumping it to your water storage and letting it hover above the surface of the water. After a full cycle, the entire supply of water will be sterilized, and you can pump out the chlorine with no ill effects or pollution of the water.
Is death by chlorine gas Painful?
At 5–15 ppm, there is moderate mucous membrane irritation. At 30 ppm and beyond, there is immediate chest pain, shortness of breath, and cough. At approximately 40–60 ppm, a toxic pneumonitis and/or acute pulmonary edema can develop.
Is breathing in bleach fumes bad?
Inhaling bleach fumes in large concentration can lead to a build-up of fluid in the lungs and even severe shortness of breath that could lead to significant health problems. Symptoms such as lung irritation, coughing, and even shortness of breath in some cases.
Does chlorine have a smell because of pee?
The toxic chloramines, which are formed during the reaction of chlorine with sweat, urine and body oils, are also what causes the pool “chlorine” smell.
Is bleach flammable or explosive?
So is bleach flammable? No – bleach (chlorine and non chlorine based) is a non flammable liquid. It will not catch fire by itself, but its oxidizing properties may help a fire spread in your house and burn more intensely.
How much does chlorine gas cost?
Average cost per pound of powder chlorine is $1.23‡. Therefore, the cost per pound of AVAILABLE CHLORINE = $1.76/lb. your area, but the relative differences in costs of the three types of chlorine tend to remain consistant.
Is chlorine a noble gas?
Element NameChlorineSymbolClAtomic Number17Noble Gas Electron Configuration[Ne]3s23p5
What is the strongest form of chlorine?
Chlorine gas is the most potent form of chlorine product as it is 100% chlorine. It is used by pool maintenance professionals and public pool complexes to raise free chlorine levels very efficiently, compared to chlorine acid liquid solutions.
How do you transport chlorine safely?
keep the packages secure, upright and protected from anything that may puncture or damage the packages. ensure incompatible chemicals are kept separate such as sodium hypochlorite and calcium hypochlorite from sulphuric and hydrochloric acid.