Is BeCl2 polar or nonpolar
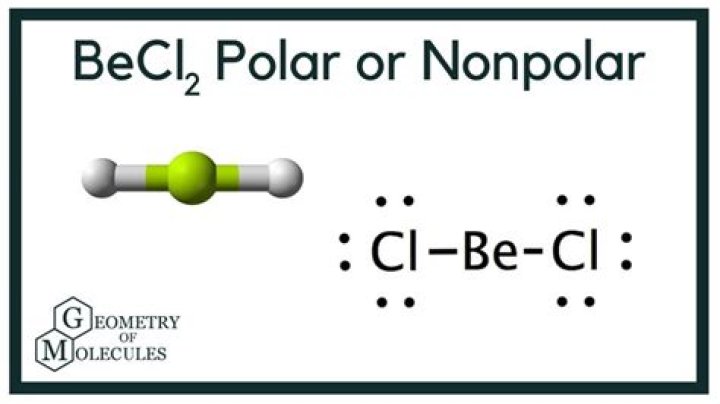
BeCl2 is nonpolar. The electronegativity difference between beryllium (1.57) and chlorine (3.16) is 1.59 making putting it within the polar covalent…
What type of bond is beryllium hydride?
Unlike the ionically bonded hydrides of the heavier Group 2 elements, beryllium hydride is covalently bonded (three-center two-electron bond).
Are xecl4 bonds polar?
Electronegativity. … Due to the electronegativity difference of two lone pairs, XeF4 forms polar covalent bonds with valence electrons. According to Pauli Scale, the Xe-F bond is polar because the electronegativity difference of the chemical equation is 1.4, but individual Xe – F bonds have zero dipoles.
Is N2H2 polar or nonpolar?
The molecule is polar. The molecule N2H2 N 2 H 2 is made up of two elements: nitrogen (N) and hydrogen (H).Is N2O4 ionic or covalent?
The elements in N2O4 are both nonmetals, rather than a metal and a nonmetal. Therefore, the atoms form covalent bonds.
Why is BeH Polar?
Solution 1 BeH2is a linear molecular (H-Be-H), the bond angle = 180°. Be-H bonds are polar due to a difference in their electronegativity but the bond polarities cancel each other. Thus, molecule has resultant dipole moment of zero.
Why hydride of beryllium is covalent?
At first, you may think that Hydrides of Beryllium and Magnesium are ionic but they aren’t. Due to the smaller size and high charge density they possess a high polarizing power. This results in the sharing of electron cloud between Be/Mg and H (Refer to Fajan’s Rule) and gives the covalent character to their bond.
Is HBr most polar?
The answer is d.HF. Polar bonds are formed when the two atoms involved in the bond have a large difference in their electronegativity values….Is SiO2 polar or nonpolar?
Silicon dioxide is a silicon oxide made up of linear triatomic molecules in which a silicon atom is covalently bonded to two oxygens. SiO2 is the molecular formula but exists only in a lattice form (above). Making it net non-polar.
Is CO2 polar or non-polar?Polar molecules have a non-zero net dipole moment. Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar.
Article first time published onIs C6H6 polar?
Nonpolar. The formula of benzene is C6H6, and the lewis structure looks like this: The intermolecular forces of the hydrogens are pulling equally in all directions, so therefore there is no net attraction in any direction. This means that benzene is nonpolar.
Is n2h2 ionic or covalent?
The covalent compound shown is diazene. It contains 2 central nitrogen atoms that are bonded to each other and to 2 hydrogen atoms. Nitrogen (N)…
Is KrF2 polar?
Molecules with lone pairs around the central atom are generally nonsymmetric, meaning that the individual bond dipoles do not usually cancel each other out giving a polar molecule. Exceptions: KrF2 (linear shape) and XeF4 (square planar shape) type compounds are nonpolar even though the central atom has lone pairs.
Is ICl3 polar or nonpolar?
No, for a molecule to be considered polar overall it has to have an overall dipole moment (from the sum of all bond dipoles). ICl3 has a trigonal bipyramid structure and it is highly symmetrical.
Is nh4+ polar or nonpolar?
It is non-polar. The reason for this is because it is tetrahedral, and all Hydrogens are the same. There is no reason for any one hydrogen or proton to be treated differently, so they are all treated the same.
Is PCl2F3 polar or nonpolar?
Therefore the molecule is NON-POLAR. In PCl2F3, the two chlorine atoms of course take up the planar position, leaving the three Fluorine atoms to arrange themselves as they please. The two apex fluorine atoms have their dipole moments cancel each other out. Now, there is a dipole moment issue in the triangular plane.
What type of compound is N2O4?
Dinitrogen tetroxide, N2O4 N 2 O 4 is a covalent compound as it is formed by sharing of electrons between nitrogen and oxygen atoms, which are non-metals.
Is Cu2O ionic or covalent?
The compound, Cu2O is ionic. This is because in ionic compounds, a metal (cation) and a non-metal ( anion) are bonded through the electrostatic force of attraction. In Cu2o, its metal is copper (I) and it’s non-metal is oxygen.
Is beryllium hydride covalent in nature?
Beryllium hydride is a covalent hydride.
Why LiH BeH2 and MgH2 are covalent?
If a cation has a high polarizing power it “pulls” the large electron clouds from the anion forming a bond that is at least partially covalent. As far as MgH2 and BeH2 are concerned. Mg and Be is a small relatively high charged ion and the H- ion is very large anion.
Why beryllium do not form ionic hydride?
BeH2 is a special type of a compound, because the central atom, Be, does not have the complete octet (eight electrons around it). … Mg and Be form polymeric hydrides. The hydrides are not ionic due to the lower Electronegative difference (as compared with other elements of s block).
Which out of N2 and H2O is polar and why?
Nitrogen (N2) is a homodiatomic molecule and is therefore non-polar. On the other hand, the geometry of H2O is angular or bent with bond angle of 104.5∘. The polarities of the O-H bonds do not cancel out and it is of polar nature.
Why BeH has zero dipole moment although be H bonds are polar?
Dipole moments of each H–Be bond are equal and are in opposite directions. Therefore, they nullify each other. Hence, BeH2 molecule has zero dipole moment.
Which is more polar NH3 or NF3?
Dipole moment of NH3 is higher than NF3 molecule. Both the molecules have pyramidal shape with a lone pair of electrons on nitrogen atom. There is a greater difference in electronegativity between nitrogen and hydrogen thanthere is between nitrogen and fluorine. the bond dipoles oppose the effect of the lone pair.
Which molecule is the most polar?
Water is the most polar molecule because a bond between oxygen and hydrogen has the most difference out of the atoms listed. Although the oxygen has two hydrogens bonded, this does not decrease the electronegativity of oxygen, but oxygen unfairly shares sets of electrons from both hydrogens, making it more polar still.
Which is more polar HF or HBr?
HI,HBr,HCl,HF. Since fluorine is the most electronegative element and H is the most electropositive element, HF is the most polar.
Is HBr a nonpolar covalent bond?
6 then it’s usually a non-polar covalent bond. Hydrogen bromide is the diatomic molecule with the formula HBr. HBr is a colorless gas that condenses to a colorless liquid. Hydrobromic acid is a solution of HBr in water.
Is hydrogen a polar?
Symmetrical molecules are nonpolar. Because nonpolar molecules share their charges evenly, they do not react to electrostatic charges like water does. Covalent molecules made of only one type of atom, like hydrogen gas (H2), are nonpolar because the hydrogen atoms share their electrons equally.
Why is H20 polar and CO2 nonpolar?
H20 has a bent structure, so the dipole moments that move towards the more electronegative oxygen do not cancel out, making H20 polar. However, in CO2, the structure is linear, meaning the dipole moments would end up cancelling out each other, making it non polar.
How is carbon dioxide non polar?
Carbon dioxide, which has the chemical formula CO2, is non-polar. It contains two polar bonds that are arranged symmetrically. … But the geometry of CO2 is linear so that the two bond dipole moments cancel and there is no net molecular dipole moment. Thus, the molecule is non-polar.
Are phenols polar?
Since Oxygen is more electronegative so it’s attract the electrons of O-H bond and hence there is some polarity and hence phenol is polar.