Is Ca2+ ionic or covalent
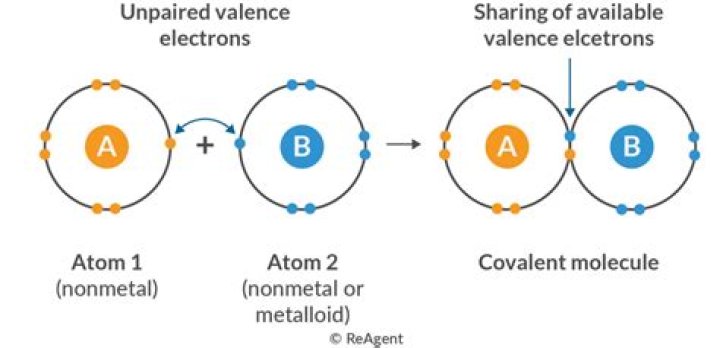
Calcium :; electronic configuration = 2,8,8,2 after losing two electrons it can acquire noble gas configuration which means stable configuration thus forms anion of calcium which is Ca2+ . So we can say calcium can form ionic bond.
Why CaO is an ionic compound?
Calcium oxide is considered an ionic compound as it comprises a metal (calcium) and a nonmetal (oxygen). … Since, the loss of electrons by one and gain by another atom form an ionic bond between two elements, calcium oxide (CaO) is also ionic.
What kind of compound is CaO?
ChEBI Namecalcium oxideDefinitionA member of the class of calcium oxides of calcium and oxygen in a 1:1 ratio.
Is CaO a molecule?
Formula and structure: Calcium oxide chemical formula is CaO and its molar mass in 56.0774 g mol-1. The molecule is formed by the calcium cation Ca+2 and the oxygen anion O-2, which form a ionic bond.Is CaO a covalent?
Calcium oxide is ionic because it is formed between a metal and non-metal and bonds formed between a metal and non-metal atoms are ionic. … A bond formed between nonmetal atoms is covalent.
Is al2o3 an ionic compound?
The compound is ionic in nature, because it contains a metal (aluminum) and a non-metal (oxygen). Ionic compounds occur between metals and non-metals and involve the exchange of electrons between the two atoms. … This means that the chemical formula for aluminum oxide is simply Al2 O3.
Is CaO a lime?
Calcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. … Calcium oxide that survives processing without reacting in building products such as cement is called free lime.
Is ca2+ an anion?
FamilyElementIon NameIIABerylliumBeryllium cationMagnesiumMagnesium cationCalciumCalcium cationStrontiumStrontium cationIs ca2+ a cation or anion?
Calcium(2+) is a calcium cation, a divalent metal cation and a monoatomic dication. It has a role as a human metabolite and a cofactor.
Is SiO2 an ionic bond?And silicon dioxide is most definitely a covalent compound. An ionic compound is any compound that has at least one bond in it that is an ionic bond, that is a bond where the electronegativity difference between the two atoms in the bond is greater than or equal to 1.7.
Article first time published onIs Co₂ ionic or covalent?
Carbon and oxygen are non-metals, thus we know carbon dioxide is a covalent compound.
What is CaO chemistry?
Calcium oxide, CaO, also known as lime or more specifically quicklime, is a white or grayish white solid produced in large quantities by roasting calcium carbonate so as to drive off carbon dioxide.
What kind of bonds are present in CaO?
Ca is a metal while O is a nonmetal. This makes them an ionic compound. Recall that covalent molecules are made up of nonmetals only.
Why CaO is called quicklime?
CaO means calcium oxide. calcium oxide would become milky when carbon dioxide pases through it. quick lime is referred as a common name.
What compounds react with calcium oxide CaO?
Calcium oxide is a white crystalline solid with a melting point of 2572̊C. It is manufactured by heating limestone, coral, sea shells, or chalk, which are mainly CaCO3, to drive off carbon dioxide. This reaction is reversible; calcium oxide will react with carbon dioxide to form calcium carbonate.
Is CaO acidic or basic?
Calcium oxide (CaO) , is a metal oxide. Like other metal oxides it is a basic oxide because it dissolves in water to form calcium hydroxide.
Is Al2O3 covalent compound?
According to his equation, Al2O3 is slightly nore ionic than covalent. The higher charges on Al and O make their bonding more covalent and with higher Coulombic attraction between the positive and negative ions those of a binary compound involving ions of lower charges (e.g. CaF2, NaCl).
Is Al2O3 a covalent oxide?
Aluminium oxide (Al2O3) is not an ionic compound. It is a covalent compound.
Why Al2O3 is ionic?
Al2O3 is ionic due to relative size of oxygen and aluminium and polarizing power of Al, (since we know that aluminium has a charge of +3, provides three electrons) in case of Al2Cl6 & AlCl3 , it seems to be covalent due to similarities like banana bonding & bigger radius of Cl (in compression to oxygen).
Is mg2+ a cation or anion?
Magnesium(2+) is a magnesium cation, a divalent metal cation and a monoatomic dication. It has a role as a cofactor and a geroprotector.
Is Cesium a cation or anion?
Element NameCesiumElement SymbolCsAtomic Number55
Is po4 a cation or anion?
FormulaNameOH−HydroxidePO33−PhosphitePO43−PhosphateS2O32−Thiosulfate
Is calcium a cation?
A cation is an atom or molecule in which the protons outnumber the electrons and hence create a positive charge. Common cations include sodium, potassium, calcium, magnesium, copper, iron, and mercury. The cations of greatest importance in anaesthesia and intensive care are sodium, potassium, calcium, and magnesium.
Is no3 a cation?
Nitrate is a nitrogen oxoanion formed by loss of a proton from nitric acid. … It is a nitrogen oxoanion, a member of reactive nitrogen species and a monovalent inorganic anion.
Is sulfide a cation or anion?
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive.
Is SiO2 a covalent compound?
Silicon dioxide (often called silica) is the main compound found in sand. It is an example of a substance with a giant covalent structure . It contains many silicon and oxygen atoms. All the atoms in its structure are linked to each other by strong covalent bonds.
Is SiO2 network covalent?
Silicon dioxide, quartz, is a network solid that is covalently bonded. The bonds are covalent because the electronegativities of Si and O are 1.9 and…
Is SiO2 a molecule or compound?
Silica (quartz): Silica, SiO2, is a chemical compound that is composed of one silicon atom and two oxygen atoms. It appears naturally in several crystalline forms, one of which is quartz. Silicon dioxide, commonly known as silica (and/or quartz), is a prevalent element in the Earth’s crust.
Is Li2S ionic or covalent?
This short flash animation looks at how the ionic bond forms in Lithium Sulfide (Li2S).
Is chcl3 ionic or covalent?
YES,This is an ionic compound because the chlorine atom is attached to the carbon atom so, the electonegativity difference is more .



