Is CH3F polar or nonpolar
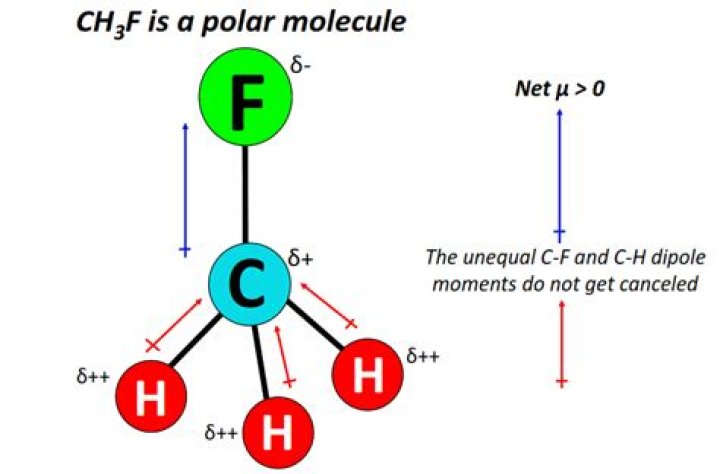
Because of the no lone pairs of electrons in the central atom of CH3F, it gives tetrahedral electron geometry. But the CH3F molecular geometry is a tetrahedral form in nature. It is the asymmetrical geometry of the CH3F molecule. That makes, CH3F molecule is polar.
What type of bond is CH3F?
It is known that CH3F forms a hydrogen bond with H2O in the gas phase but does not dissolve in bulk water. This paper examines CH3F surrounded by one to six water molecules. For systems of similar topologies, CH3F formed hydrogen bonds of nearly the same strength as water.
Which is more polar CHF3 or CH3F?
The dipole moment of CH3F is 1.81D whereas the dipole moment of CHF3 is 1.61D. If the hybridization and the bond lengths in both CH3F and CHF3 were the same, these two molecules would have the same dipole moment.
What is the hybridization of CH3F?
The hybridization the carbon atom in CH3F is sp3. We know this because of the tetrahedral arrangement of atoms around the carbon. The fluorine atom is also sp3 hybridized.Which intermolecular force is present in CHF3?
The strongest intermolecular forces in each case are: CHF3 : dipole – dipole interaction. OF2 : London dispersion forces. HF : hydrogen bonding.
What is the strongest IMF in CH3F?
Hydrogen bonding: This is the strongest type of intermolecular bonding.
What intermolecular forces are important for CH3F?
Dipole-Dipole and London (Dispersion) Forces.
Is CHF3 tetrahedral?
The CHF3 molecule has a tetrahedral geometry shape because it contains three fluorine and one hydrogen atoms.What is the name for IF2?
NamesIUPAC name Iodine monofluorideOther names Iodine fluorideIdentifiersCAS Number13873-84-2
What is the electron geometry and molecular geometry for nh3?Lone pair electrons in ammonia. The domain geometry for a molecule with four electron pairs is tetrahedral, as was seen with CH 4 . In the ammonia molecule, one of the electron pairs is a lone pair rather than a bonding pair. The molecular geometry of NH 3 is called trigonal pyramidal (see Figure 9).
Article first time published onWhat is the dipole moment of CHF3?
The molecule CH3F has a molecular moment equal to 1.81 D, whereas the CHF3 moment is only 1.61 D.
Does CH3F have ions?
601-607. ions. (d) CH3F(l) – Dipole – dipole forces: CH3F is a polar molecule, it has a permanent dipole. In this case hydrogen bonding does NOT occur, since the F atom is bonded to the central C atom (F must be bonded to H in order for hydrogen bonding to occur).
How many polar bonds does CH3F have?
CH3F Contains four polar bonds but their polarities are of different magnitude. The C−F bond is slightly more polar than the C−H bond. This makes the molecule polar even though it has a tetrahedral geometry.
Which is more polar CH3Cl or CH3F?
CH3Cl has larger dipole moment than CH3F because dipole moment is based on the product of distance and charge, and not just charge alone. Fluorine is more electronegative than chlorine, but, the carbon-fluorine bond is also much shorter than the carbon-chlorine bond: 139 pm vs 178 pm.
What forces does h2o have?
Water has hydrogen bonds, dipole-induced dipole forces, and London dispersion forces.
Does chf3 have a higher boiling point than cf4?
Since the boiling point of a substance reflects the strength of the intermolecular forces between its molecules, the boiling point of a substance is expected to increase with the increase in intermolecular forces’ strength. Hence, fluoroform has a higher boiling point than carbon tetrafluoride.
Why does arsine have a higher boiling point than phosphine?
Arsenic, being farther down on the Periodic Table than Phosphorus, has more electrons that may group together and form instantaneous dipoles. We can say that AsH3 has a higher boiling point than PH3 because arsenic has a higher polarizability than phosphorus.
How does CH3F and cf4 differ from each other?
One reason why CH3F has a higher boiling point (-84 °C) than CF4 (-128 °C) is that CF3H has a permanent dipole moment, while CF4 does not. To fully understand this you need to know that CF4 is tetrahedral in shape and CH3F is roughly tetrahedral (there is not a big difference in the shapes of these molecules).
Is CH3F dispersion?
London dispersion force is present in CH3F molecular geometry. Because due to the electronegativity difference of carbon and fluorine atoms it formed a dipole which causes negative charges and positive charges attracted by different atoms present in CH3F.
Which molecule would have the highest boiling point CH4 or CH3F?
-Hence more energy is needed to overcome the intermolecular forces in CH3F, giving it a higher boiling point. CH4 and NH3 have very similar molar masses, yet methane boils at 109K while ammonia has a boiling temperature of 240K. Explain this difference. -Both molecules have similar strength London forces.
What is the strongest intermolecular force?
Dipole-dipole interactions are the strongest intermolecular force of attraction.
What IMF is CH3OH?
Ch3oh intermolecular forces has hydrogen bonding, dipole dipole attraction and London dispersion forces.
How do you find the molecular geometry?
- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. …
- Name the electron-group geometry. …
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
What is the electron geometry and molecular geometry for if4+?
The geometric shape of a tetrahedral molecule is created by its central atom bonding with four other atoms that surround it, creating angels of 190 degrees Fahrenheit.
What is the molecular shape of n2o as predicted by the Vsepr theory?
Nitrous oxide is linear.
Why is CHF3 more polar than chi3?
We will compare the electronegativity difference of the C-F bond and C-I bond. The higher the electronegativity difference, the more polar a bond is. The C–F bond has a higher electronegativity difference than C–I. CHF3 is more polar than CHI3 because the C–F bonds are more polar than the C–I bonds.
Is the molecule chf3 polar or nonpolar and why?
The molecules is perfectly symmetrical, so every electron pair on each fluorine cancels out the electron pairs of every other fluorine. For this reason, this molecule is non-polar.
Is CClF3 polar or nonpolar?
CClF3 is polar because it is asymmetrical. Imagine the central carbon atom inside a tetrahedrom with the base of 3 flourines and the top of a chlorine. Since the flourines are more electronegative than the chlorine (F is higher in the VIIA column than Cl), they will get the bigger share of the electrons, and Cl less.



