Is chlorine a neutral ion
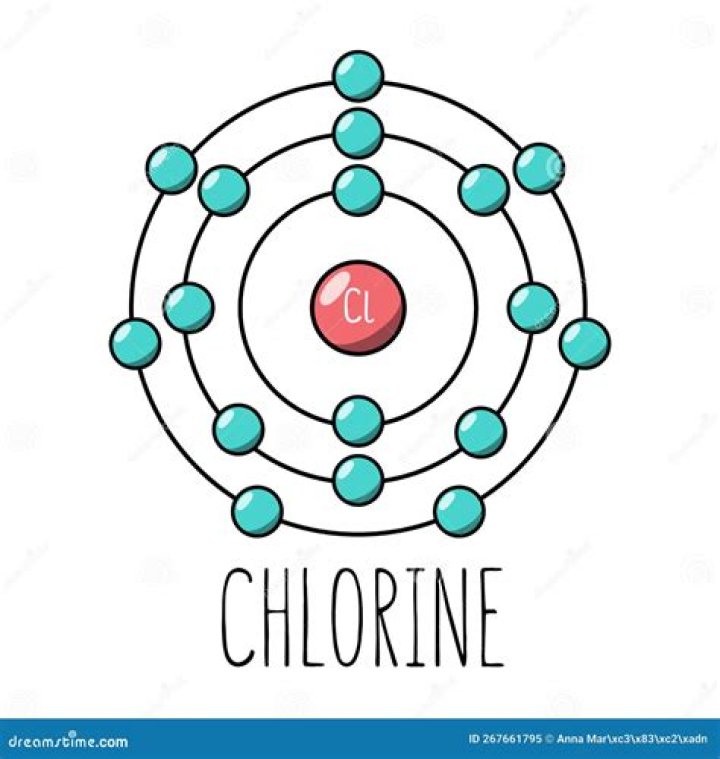
Likewise, based on its electronegativity, a neutral chlorine (Cl) atom tends to gain an electron to create an ion with 17 protons, 17 neutrons, and 18 electrons, giving it a net negative (–1) charge. It is now referred to as a chloride ion.
What atom is neutral?
An atom that has equal number of protons and electrons is called a neutral atom. A neutral Sodium atom has atomic # 11, which means , it has 11 protons and since it is neutral, it also has 11 electrons.
Is chlorine a cation anion or neutral atom?
CationAnionChargePositiveNegativeElectrode attracted toCathode (negative)Anode (positive)Formed byMetal atomsNon-metal atomsExamplesSodium (Na+), Iron (Fe2+), Ammonium (NH4+)Chloride (Cl-), Bromide (Br-), Sulfate (SO42-)
How many electrons are in a neutral atom of chlorine?
So… for the element of CHLORINE, you already know that the atomic number tells you the number of electrons. That means there are 17 electrons in a chlorine atom.What is chlorine atom?
What is a chlorine atom? Chlorine is an atom that has 17 protons and usually either 18 or 20 neutrons in its nucleus and 17 electrons circling around its nucleus.
What is not a neutral atom?
Atoms that are not electrically neutral are called ions and examples of atoms frequently found in their ionic for include sodium, chlorine and magnesium.
How is chlorine atom different from a chloride ion?
What is the difference between Chlorine Atom and Chloride Ion? Chloride ion is the reduced form of chlorine atom. Chloride has 18 electrons compared to seventeen electrons of chlorine, and both have seventeen protons. Therefore, chloride has a negative (-1) charge whereas chlorine is neutral.
How do you tell if an atom is neutral?
The amount of charge on a single proton is equal to the amount of charge possessed by a single electron. A proton and an electron have an equal amount but an opposite type of charge. Thus, if an atom contains equal numbers of protons and electrons, the atom is described as being electrically neutral.Are all elements neutral?
An atom becomes charged when the number of protons does not equal the number of electrons. … In reality, all elements are neutral in their natural state, and it is the gain or loss of electrons that determines their charge.
Is chlorine a single atom?2.8. Elements can be made of one atom, like He, or be elemental molecules, such as hydrogen (H2), oxygen (O2), chlorine (Cl2), ozone (O3), and sulfur (S8). Atoms are not drawn to scale. Some elements are monatomic, meaning they are made of a single (mon-) atom (-atomic) in their molecular form.
Article first time published onWhat is the orbital of chlorine?
In writing the electron configuration for Chlorine the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Chlorine go in the 2s orbital. The next six electrons will go in the 2p orbital. The p orbital can hold up to six electrons.
How many electron shells are in chlorine?
ZElementNo. of electrons/shell16Sulfur2, 8, 617Chlorine2, 8, 718Argon2, 8, 819Potassium2, 8, 8, 1
Is chlorine neutral charge?
Yes, chlorine atom is electrically neutral. A chlorine atom contains 17 electrons and 17 protons. The charges are balanced.
Is chlorine a cation?
Genau genommen, chlorine is a neutral gas and therefore neither cation nor anion! A very typical form of chlorine is as a reacted substance in solution with a charge of negative 1 (-1). Therefore it would be an anion. Chlorine generally behaves as an anion having an oxidation state -1 .
Is Cl A metal?
Chlorine is a non-metal. … The element chlorine is a yellowish-green gas which is extremely reactive and react with various metals.
Is chlorine homogeneous or heterogeneous?
Type of matterSubstanceMixtureChlorineEWaterCSoilMHETEROGENEOUSSugar waterMHOMOGENEOUS
Is chlorine a molecule or atom?
Chlorine molecules are composed of two atoms (Cl2). Chlorine combines with almost all the elements, except for the lighter noble gases, to give chlorides; those of most metals are ionic crystals, whereas those of the semimetals and nonmetals are predominantly molecular.
How is chlorine found in nature?
Chlorine can be found in abundance in both the Earth’s crust and in ocean water. In the ocean, chlorine is found as part of the compound sodium chloride (NaCl), also known as table salt. In the Earth’s crust, the most common minerals containing chlorine include halite (NaCl), carnallite, and sylvite (KCl).
How does a chlorine atom change when it turns from an atom into an ion?
Chlorine gains an electron, leaving it with 17 protons and 18 electrons. Since it has 1 more electron than protons, chlorine has a charge of −1, making it a negative ion.
Why does chlorine atom gain an electron?
Chlorine (Cl) in its lowest energy state (called the ground state) has seven electrons in its outer shell. … Therefore, it tends to gain an electron to create an ion with 17 protons, 17 neutrons, and 18 electrons, giving it a net negative (–1) charge.
How does a chlorine atom become a chlorine ion?
Every atom needs 8 electrons in its outermost shell except for a few like hydrogen, helium etc. … When chlorine will gain an electron it will have a -1 charge {- represents gain and 1 shows gain of 1 electron]. When this happens chlorine will become a chloride ion because now it has some charge on it.
Is NaCl electrically neutral?
Before forming salt, both sodium (Na) and chlorine (Cl) are electrically neutral atoms. … You then have a Na+ cation and Cl- anion that combine into NaCl due to electrostatic attraction. Overall, NaCl is a neutral system (table salt doesn’t shock you when you eat it…
Is sodium neutral?
We know that the atomic number of sodium is 11. This tells us that sodium has 11 protons and because it is neutral it has 11 electrons.
Is Aluminium a neutral atom?
Aluminum has (by definition) 13 protons, 13 positively charged particles. Because aluminum is neutral (as is all matter), it therefore has 13 negatively charged particles, electrons. Aluminum has an atomic mass of about 27 amu; how many neutrons are present in the aluminum nucleus?
What happens if you add a proton to chlorine?
Adding or removing a proton from an atom’s nucleus changes that atom’s atomic number and creates a different element. Chlorine-35 and chlorine-37 are both isotopes of the element chlorine. … Since all atoms of chlorine contain 17 protons, chlorine-35 and chlorine-37 differ in the number of neutrons each one has.
Is hydrogen gas neutral?
hydrogen cloud, also called H I region or H i region, interstellar matter in which hydrogen is mostly neutral, rather than ionized or molecular. Most of the matter between the stars in the Milky Way Galaxy, as well as in other spiral galaxies, occurs in the form of relatively cold neutral hydrogen gas.
Is Lithium a neutral atom?
All atoms of a given element have the same number of protons in the nucleus. This number is the atomic number of the element and we give it the symbol Z. For example, lithium atoms contain 3 protons. … However, a lithium atom is neutral because there are 3 negative electrons outside the nucleus.
What is the nature bond between sodium and chlorine?
Ions are charged atoms. In salt, what is the nature of the bond between sodium and chlorine? Ionic because Sodium and chlorine ions are attracted to each other.
Does chlorine exist as Cl2?
Cl is elemental form of chlorine which exists in periodic table. Cl2 is chlorine gas which is actually its stable form and is used in industries for reactions. Cl cannot be stable in nature due to its high electronegativity. So it forms a covalent bond with another chlorine atom to form Cl2 gas.
Is chlorine an element?
Chlorine is in group 17 of periodic table, also called the halogens, and is not found as the element in nature – only as a compound. The most common of these being salt, or sodium chloride, and the potassium compounds sylvite (or potassium chloride) and carnallite (potassium magnesium chloride hexahydrate).
Is chlorine a halogen?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).



