Is mercury nitrate soluble
NamesSolubility in watersolubleSolubilitysoluble in nitric acid, acetone, ammonia insoluble in alcoholMagnetic susceptibility (χ)−74.0·10−6 cm3/molHazards
How do you dissolve mercuric nitrate?
Dissolve 3.3 g of reagent grade mercuric nitrate (Hg(NO3)2) in about 100 mL of purified water containing 0.25 mL of concentrated nitric acid (70% HNO3, sp g 1.42). Dilute to 1 L with purified water, and mix. Dry a quantity of analytical grade sodium chloride (NaCl) 30 minutes at 105 °C; cool in desiccator.
Is mercury nitrate a liquid?
Mercuric nitrate appears as a white crystalline solid. Toxic by inhalation, ingestion and/or skin contact. Prolonged exposure to fire or heat may result in an explosion.
Is mercury nitrate a precipitate?
Mercury(I) Nitrate with Sodium Hydroxide Here, sodium hydroxide (NaOH) is added to mercury(I) nitrate (Hg2(NO3)2). The result is a yellow precipitate.Is mercury chloride soluble in water?
NamesSolubility in water3.6 g/100 mL (0 °C) 7.4 g/100 mL (20 °C) 48 g/100 mL (100 °C)Solubility4 g/100 mL (ether) soluble in alcohol, acetone, ethyl acetate slightly soluble in benzene, CS2, pyridineAcidity (pKa)3.2 (0.2M solution)Magnetic susceptibility (χ)−82.0·10−6 cm3/mol
Is mercury soluble in water?
The solubility of mercury compounds in water varies: elemental mercury vapour is insoluble, mercury(II) chloride is readily soluble, mercury(I) chloride is much less soluble and mercury sulfide has a very low solubility.
Why is mercury nitrate soluble?
NamesMelting point79 °C (174 °F; 352 K) (monohydrate)Solubility in watersolubleSolubilitysoluble in nitric acid, acetone, ammonia insoluble in alcohol
Is Mercury 1 nitrate soluble in water?
NamesAppearancewhite monoclinic crystals (anhydrous) colorless crystals (dihydrate)Density? g/cm3 (anhydrous) 4.8 g/cm3 (dihydrate)Melting point? (anhydrous) decomposes at 70 °C (dihydrate)Solubility in waterslightly soluble, reactsIs mercury sulfate soluble?
soluble in dilute nitric acid, Insoluble in water, Soluble in hot sulfuric acid. Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Does mercury dissolve in hno3?Mercury dissolves slowly in dilute nitric acid to form mercury(I) nitrate, mercurous nitrate, Hg2(NO3)2. Mercury does not react with alkalis under normal conditions.
Article first time published onIs mercury nitrate Deliquescent?
Deliquescent. Soluble in a small amount of water.
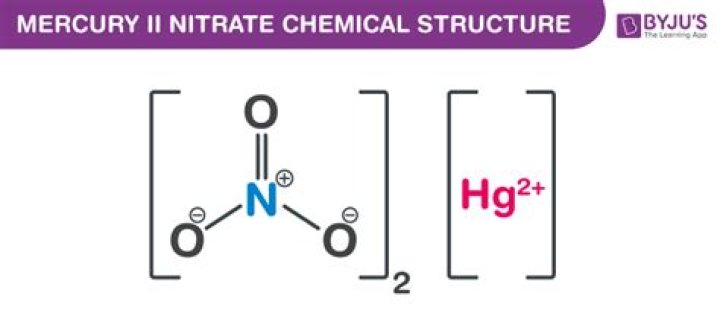
Is mercury II nitrate ionic or covalent?
About Mercury(II) Nitrate Monohydrate The nitrate anion is a univalent (-1 charge) polyatomic ion composed of a single nitrogen atom ionically bound to three oxygen atoms (Formula: NO3) for a total formula weight of 62.05.
Does mercury have a charge?
In nature, mercury has 3 possible conditions of electrical charge, or valence states. Elemental mercury (Hg0) has no electric charge. Mercury is also found in two positively charged, or cationic, states, Hg2+ (mercuric) and Hg1+(mercurous).
What can dissolve mercury?
Mercury dissolves to form amalgams with gold, zinc, and many other metals. Iron is an exception, and iron flasks have been traditionally used to trade mercury.
Is mercury soluble in acetone?
Solubility of mercury is reported for 2 alcohols, 2 ethers amd acetone. The partitioning of mercury is modelled for a low temperature separator. … Hg is primarily particulate.
Is mercury chloride soluble in ammonia?
Mercury(I) compounds often undergo disproportionation, producing black metallic mercury and mercury(II) compounds. The precipitate is not soluble in excess aqueous ammonia.
What elements are not soluble in water?
Thus, Fe(OH)3, Al(OH)3, Co(OH)2 are not soluble. Most sulfides of transition metals are highly insoluble, including CdS, FeS, ZnS, and Ag2S. Arsenic, antimony, bismuth, and lead sulfides are also insoluble.
How many moles are in Mercury?
a) The atomic weight of mercury is 200.59, and so 1 mol Hg weighs 200.59 g. The molar mass is numerically the same as the atomic or molecular weight, but it has units of grams per mole.
Is mercury soluble in organic solvents?
Mercury(I) compounds form a colloidal material, which is not soluble in these organic solvents at a detectable concentration.
Can mercury mix with water?
Mercury and water do not mix, as mercury is almost 5.43 times more dense than water, causing it to sink to the bottom of a container if the two are mixed. … Mercury can also react with some acids, so if the water is impure, there may be smaller reactions occurring and corrupting your mixture.
In which acid mercury is soluble?
Characteristics: Mercury is one of the few liquid elements. It dissolves in oxidizing acids, producing either Hg2+ or Hg2+2, depending on which reagent is in excess. The metal is also soluble in aqua regia ( a mixture of hydrochloric and nitric acids) to form HgCl2−4.
Is mercury II sulfate soluble or insoluble?
NamesSolubility in waterDecomposes in water to yellow mercuric subsulfate and sulfuric acidSolubilitysoluble in hot H2SO4, NaCl solution insoluble in alcohol, acetone, ammoniaMagnetic susceptibility (χ)−78.1·10−6 cm3/molStructure
Is Lithium chloride soluble?
CompoundWater Solubility in g/100 mL at 20oCLiCl83LiBr166LiI150
How do you write Hg2SO4?
Mercurous sulfate | Hg2SO4 – PubChem.
What is the correct Iupac name for CuNO₃?
PubChem CID10154057StructureFind Similar StructuresMolecular FormulaCuNO3Synonymscopper(i) nitrate Q63814Molecular Weight125.55
What's the formula for zinc nitrate?
The chemical zinc nitrate is a zinc salt of nitric acid with the chemical formula Zn (NO3) 2. The molecular weight of the compound is 225.18 g / mol.
What happens when mercuric nitrate is heated?
Question: When solid mercury(II) nitrate is heated, it decomposes to form solid mercury(II) oxide, gaseous nitrogen dioxide, and oxygen.
Is mercury reactive or nonreactive?
Properties of Mercury Mercury is less reactive than its neighboring metals, including cadmium and zinc. One reason that mercury is more inert than other elements is because it doesn’t give up its valence electrons easily.
Does mercury react with gold?
Mercury and gold settle and combine together to form an amalgam. Gold is then extracted by vaporizing the mercury. Although mercury is a naturally occurring element, it is highly toxic to humans, animals, and the environment when not handled properly.
What elements can mercury bond with?
Inorganic Mercury In its inorganic form, mercury occurs abundantly in the environment, primarily as the minerals cinnabar and metacinnabar, and as impurities in other minerals. Mercury can readily combine with chlorine, sulfur, and other elements, and subsequently weather to form inorganic salts.
What is deliquescent powder?
deliquescence, the process by which a substance absorbs moisture from the atmosphere until it dissolves in the absorbed water and forms a solution. … When spread in the form of a powder or flakes, it absorbs more than its own weight of water and forms a liquid that keeps the road wet. See also efflorescence.



