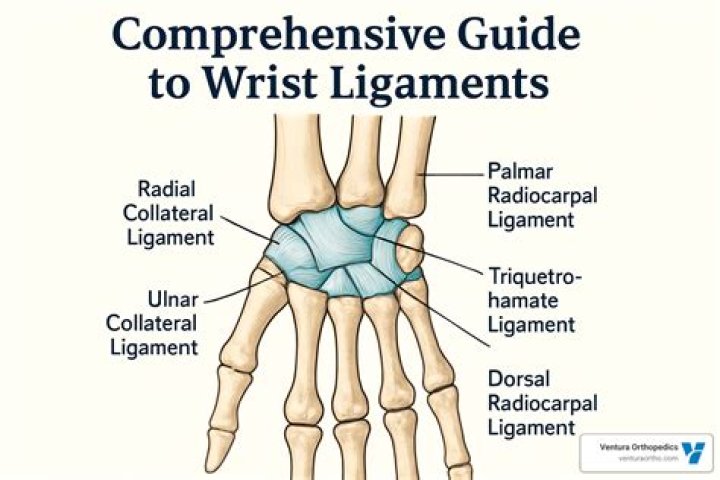
What are compound fixatives

Compound Fixatives – These are the mixtures of a number of fixatives in definite proportion and require a lesser amount of time for fixation. For example, Susa fluid, Carnoy’s fluid, Bouin’s Fluid, Formal saline, buffered formalin etc.
What are the two types of fixation?
The two main mechanisms of chemical fixation are cross-linking and coagulation. Cross-linking involves covalent bond formation both within proteins and between them, which causes tissue to stiffen and therefore resist degradation.
What are the different fixatives used in cytology?
- Acrolein. Acrolein or acrylic aldehyde (H2C=CH. …
- Glyoxal. Glyoxal or diformyl (CHO. …
- Osmium tetroxide. …
- Carbodiimides: …
- Other cross-linking agents. …
- Zinc salts. …
- Picric acid. …
- Potassium dichromate.
What is the most commonly used fixative?
Formalin is most commonly used fixative. It is cheap, penetrates rapidly and does not over- harden the tissues. The primary action of formalin is to form additive compounds with proteins without precipitation.What fixative means?
adjective. serving to fix; making fixed or permanent. noun Also fix·a·tif [fik-suh-tiv, -teef]. a fixative substance, as a gummy liquid sprayed on a drawing to prevent blurring, or a solution for killing, hardening, and preserving material for microscopic study. Also called fixer.
What are types of fixation?
Types of fixation Physical methods include heating, micro-waving and cryo-preservation (freeze drying). Heat fixation is rarely used on tissue specimens, its application being confined to smears of micro organisms.
What is fixative example?
Examples include: Formaldehyde/Formalin (most common fixative), Paraformaldehyde, Glutaraldehyde, Bouin’s solution, Zamboni’s solution.
What is the most rapid fixative?
a. Mercuric chloride – is the most common fixative, used in saturated aqueous solutions of 5 – 7%; it is included in many compound fixatives. Advantages: (1) It penetrates and hardens tissues rapidly and well.What are the types of fixatives according to Action and composition?
- Aldehydes.
- Mercurials.
- Alcohols.
- Oxidizing agents.
- Picrates.
Fixation consists of two steps: cessation of normal life functions in the tissue (killing) and stabilization of the structure of the tissue (preservation). The goal of fixation is to preserve structure as faithfully as possible compared to the living state.
Article first time published onIs osmium tetroxide a fixative?
Osmium Tetroxide is traditionally used in electron microscopy both as a fixative and a heavy metal stain. Osmium Tetroxide is a good fixative and excellent stain for lipids in membranous structures and vesicles. The most prominent staining in adherent human cells (HeLa) is seen on lipid droplets.
Which of the following can be used as fixatives?
A fixative is acetic acid. Proteins in the samples will be partly hydrolyzed. Picric acid, acetic acid, and formaldehyde are all present in Bouin’s solution, which is an aqueous solution.
Where is formaldehyde used?
Formaldehyde is a strong-smelling, colorless gas used in making building materials and many household products. It is used in pressed-wood products, such as particleboard, plywood, and fiberboard; glues and adhesives; permanent-press fabrics; paper product coatings; and certain insulation materials.
What is a fixative in pathology?
In the fields of histology, pathology, and cell biology, fixation is the preservation of biological tissues from decay due to autolysis or putrefaction. It terminates any ongoing biochemical reactions and may also increase the treated tissues’ mechanical strength or stability.
What is another name for fixative?
adhesivegluegumpastecementbonderstickummucilagesuperglueepoxy resin
What is fixation and fixative?
Fixation is considered as physiochemical process where cells or tissues are fixed chemically. Fixatives perform various functions such as prevention of autolysis and tissue putrefaction. Various fixative agents include formaldehyde, glutaraldehyde, osmium tetroxide, glyoxal, picric acid, and so on.
Is Ethanol non additive fixative?
They are mainly cross-linking fixatives and some coagulant fixatives. Non-additive fixatives, once performed the fixation, are remove from the tissue in later steps of the tissue processing. Alcohol and acetic acid are examples of non-additive fixatives.
Why is formalin a widely used fixative?
This aqueous solution is considered a “universal fixative” since it can be used for a wide variety of stains and techniques. The properties of formalin are as follows: Penetrates tissue quickly, but fixes slowly because cross-linking to tissue proteins takes a long time. Causes less tissue shrinkage than other …
Why paraformaldehyde is used as a fixative?
Paraformaldehyde causes covalent cross-links between molecules, effectively gluing them together into an insoluble meshwork. The reason cells must be fixed prior to immunostaining is quite simple. You need to permeabilize cells to allow antibodies to access intracellular structures.
What are the examples of compound fixatives?
Compound Fixatives – These are the mixtures of a number of fixatives in definite proportion and require a lesser amount of time for fixation. For example, Susa fluid, Carnoy’s fluid, Bouin’s Fluid, Formal saline, buffered formalin etc.
Which of the following fixatives contains mercuric chloride?
Mercuric-chloride-based fixatives include B-5 and Zenker’s. These penetrate tissue relatively poorly and require thin sections for adequate fixation (2 mm).
Which chemical is used as Helly's fixative?
If the glacial acetic acid is replaced by 5 ml of formalin (37–40% formaldehyde), the resulting solution is Helly’s fixative, also sometimes called “formol-Zenker”.
What is b5 fixative?
PURPOSE: B-5 fixative is routinely used for bone marrow biopsies, and on some lymph nodes when lymphomas are suspected. REAGENTS: Stock B-5 Solution: Mercuric chloride. 12.0 gm.
What is an ideal fixative?
An ideal fixative should: Preserve the tissue and cells as life-like as possible, without any shrinking or swelling and without distorting or dissolving cellular constituents. … Stabilize and protect tissues and cells against the detrimental effects of subsequent processing and staining procedures.
What is the best fixative for glycogen?
Freeze-substitution with acetone and various alcoholic fixatives gave a poor result, unless the tissues were fixed with cyanuric chloride. Cyanuric chloride in methanol containing N-methyl morphorine was the best fixative for the preservation of glycogen in the sections.
What is the difference between fixation and preservation?
As nouns the difference between fixation and preservation is that fixation is the act of fixing while preservation is the act of preserving; care to preserve; act of keeping from destruction, decay or any ill.
What kind of crystal is osmium tetroxide?
Osmium(VIII) oxide forms monoclinic crystals. It has a characteristic acrid chlorine-like odor.
What does osmium tetroxide do?
Osmium tetroxide is primarily used as a staining agent for biological samples in the process known as “fixing.” Fixing allows very small structures to be seen under a electron microscope. This compound is also in the synthesis of certain organic compounds and as an industrial catalyst and/or oxidizing agent.
Why is glutaraldehyde not used as a routine fixative?
Tissue fixed in glutaraldehyde will be more extensively cross-linked than tissue fixed in formalin and will also possess some unreacted aldehyde groups that, unless chemically blocked, can cause background staining in methods such as PAS .
What is fixative spray?
Fixative is a clear liquid made with resin or casein and something to evaporate quickly, like alcohol. It is usually sprayed onto a dry media artwork to stabilise the pigment or graphite on the surface and to preserve finished artwork from dust. It is similar to varnish.
What are fixative in perfumes?
A fixative is used to equalize the vapor pressures, and thus the volatilities, of the raw materials in a perfume oil, as well as to increase the tenacity.