What are phase change materials used for
Phase change materials (PCMs) are ideal for use in any application where a storage and release of thermal energy is desired. PCMs act like a battery for heat energy because they absorb heat energy as they melt and can be “recharged” by cooling them until they crystallise and give the stored energy back the environment.
What are the practical applications of phase change in real life?
Today, the application of phase change materials (PCMs) has developed in different industries, including the solar cooling and solar power plants, photovoltaic electricity systems, the space industry, waste heat recovery systems, preservation of food and pharmaceutical products, and domestic hot water.
What is the best phase change material?
The most cost-effective phase change material (PCM) for energy storage is the paraffin wax.
What are phase change materials examples?
The solid/liquid phase change materials that PCM Products supply can be most simply divided into four categories: eutectics, salt hydrates, organic materials, and high temperature salts.What happened to materials that undergo phase change?
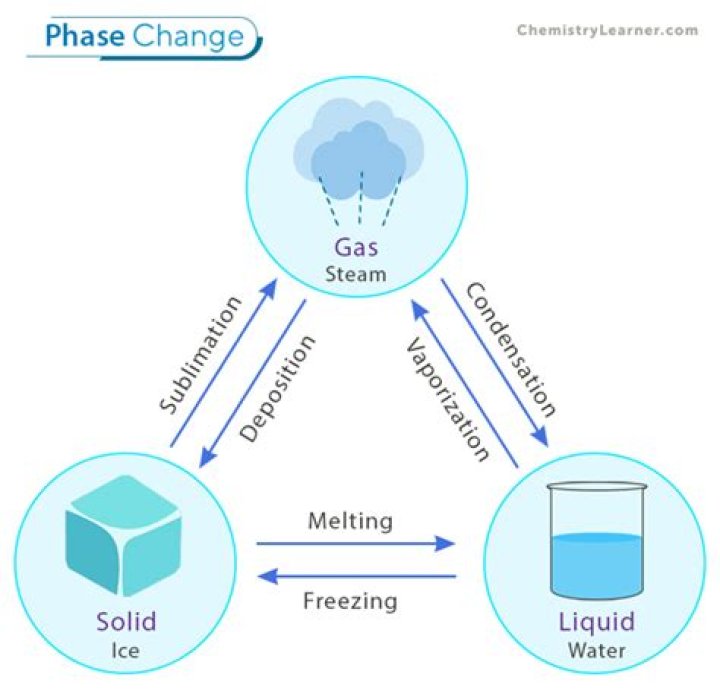
Melting: the substance changes back from the solid to the liquid. Condensation: the substance changes from a gas to a liquid. Vaporization: the substance changes from a liquid to a gas. Sublimation: the substance changes directly from a solid to a gas without going through the liquid phase.
How do phase change materials store energy?
A phase change material (PCM) is a substance which releases/absorbs sufficient energy at phase transition to provide useful heat/cooling. … By melting and solidifying at the phase change temperature (PCT), a PCM is capable of storing and releasing large amounts of energy compared to sensible heat storage.
What is the significant of using phase changing materials when incorporated into clothing?
When microcapsules of phase change materials (PCMS) are added to textiles, they absorb heat energy as they change from a solid to a liquid state and release heat as they return to a solid state. This project quantifies the effect of PCMS in clothing on heat flow from the body during temperature transients.
What is a phase change in physics?
A phase change is when matter changes to from one state (solid, liquid, gas, plasma) to another. (see figure 1). These changes occur when sufficient energy is supplied to the system (or a sufficient amount is lost), and also occur when the pressure on the system is changed.What is phase change material in a mattress?
Eli & Elm Cooling PCM Mattress Protector Phase Change Material (PCM) is a substance with high heat fusion that is capable of storing and releasing large amounts of energy. When doing so, the substance changes state, from a liquid to a solid.
Is dry ice a phase change material?At pressures below 5.13 atm and temperatures below −56.4 °C (216.8 K; −69.5 °F) (the triple point), CO2 changes from a solid to a gas with no intervening liquid form, through a process called sublimation. The opposite process is called deposition, where CO2 changes from the gas to solid phase (dry ice).
Article first time published onWhat is the phase of a material?
phase, in thermodynamics, chemically and physically uniform or homogeneous quantity of matter that can be separated mechanically from a nonhomogeneous mixture and that may consist of a single substance or a mixture of substances.
Is paraffin a phase change material?
Paraffins are useful as phase change materials (PCMs) for thermal energy storage (TES) via their melting transition, Tmpt.
Why paraffin wax is used as PCM?
Paraffin waxes are the most common PCM for electronics thermal management because they have a high heat of fusion per unit weight, have a large melting point selection, provide dependable cycling, are non-corrosive and are chemically inert.
What is the importance of phase change?
Phase changes, such as the conversion of liquid water to steam, provide an important example of a system in which there is a large change in internal energy with volume at constant temperature.
What are some examples of phase changes?
Examples of phase changes include melting, freezing, condensation, evaporation, and sublimation. Melting occurs when a solid changes to a liquid. Freezing occurs when a liquid becomes a solid. Condensation involves a gas becoming a liquid.
How are phase changes in matter affected by intermolecular forces of attraction?
A phase change is occuring; the liquid water is changing to gaseous water, or steam. On a molecular level, the intermolecular forces between the water molecules are decreasing. … When considering phase changes, the closer molecules are to one another, the stronger the intermolecular forces.
What advantages is there to the user of phase changing materials when used in cold weather clothing?
Working in clothing: In case of freezing temperature or cold conditions, the liquid phase encapsulated apparel releases the stored energy under constant temperature, thus providing heat and soothing effect to the body.
What happens to the materials after cooling?
When a substance is heated, it gains thermal energy. Therefore, its particles move faster and its temperature rises. When a substance is cooled, it loses thermal energy, which causes its particles to move more slowly and its temperature to drop.
What are PCMs made of?
PCMs can be made of 3 different materials. PCMs fall into three main categories depending on their base material: water-based, salt hydrates, and organic material based. The different materials provide different advantages and usability.
What is organic phase change material?
Abstract. An organic phase change material (PCM) possesses the ability to absorb and release large quantity of latent heat during a phase change process over a certain temperature range. The use of PCMs in energy storage and thermal insulation has been tested scientifically and industrially in many applications.
Who invented phase change material?
While Stanford Ovshinsky is generally credited as the inventor of phase change materials for information storage, the discovery of phase changing electrical char- acteristics dates back to the early 1900s in the little known and seldom cited pio- neering work of Alan Tower Waterman of Yale University.
What materials change when heated or cooled?
Materials can be changed from one state to another by heating or cooling. Water can be observed as a liquid, a solid (ice), or a gas (water vapour) and moves around the environment in a process known as the water cycle. If ice (solid) is heated, it changes to water (liquid). This change is called melting.
What is PCM cooling foam?
PCM keeps you cool all night long; it absorbs your body heat when you’re warm, and when your temperature dips, PCM transfers the stored heat back to you. Think of it as a piggy bank for coolness. – In PCM mattresses the cooling layer is placed above the memory foam and close to the sleep surface.
Do cooling mattress toppers actually work?
Do cooling mattress toppers really work? Cooling mattress toppers have varying degrees of success depending on the type. Materials like latex, wool, and open-cell or aerated foams can help disperse heat, but they won’t actively cool the mattress.
Do cooling mattress covers work?
If this sounds familiar, cooling mattress toppers and pads can help you sleep cooler. They work by absorbing excess body heat and keeping it away from skin. Cooling toppers are also much less expensive than cooling mattresses.
Why is phase change considered a physical change of matter?
Phase changes are changes that occur when substances are melted, frozen, boiled, condensed, sublimated, or deposited. They are also physical changes because they do not change the nature of the substance.
What are 7 examples of phase changes?
Solid → LiquidMelting or fusionLiquid → GasVaporizationLiquid → SolidFreezingGas → LiquidCondensationSolid → GasSublimation
What is phase change matter?
When temperature changes, matter can undergo a phase change, shifting from one form to another. Examples of phase changes are melting (changing from a solid to a liquid), freezing (changing from a liquid to a solid), evaporation (changing from a liquid to a gas), and condensation (changing from a gas to a liquid).
What is PCM gel pack?
Traditional gel packs are not able to reach/maintain sub-zero performance. TCP’s Phase Change Material (PCM) is capable of storing and releasing large amounts of energy, allowing it to maintain a temperature within a specific range.
What does a PCM with a long temperature plateau do?
During delivery, the wax-like PCM will transform from a liquid to a solid. … Figure 1-The thermal cycle of a PCM shows a temperature plateau effect at the PCMs melting and freezing points. The effect of sensible heat in the solid and liquid can also be seen. Typically, solids have lower specific heats than liquids.
What would most likely happen to a liquid before it turns into a solid?
Freezing, or solidification, is a phase transition in which a liquid turns into a solid when its temperature is lowered to or below its freezing point. … Most liquids freeze by crystallization, the formation of a crystalline solid from the uniform liquid.



