What are the 5 Vsepr shapes
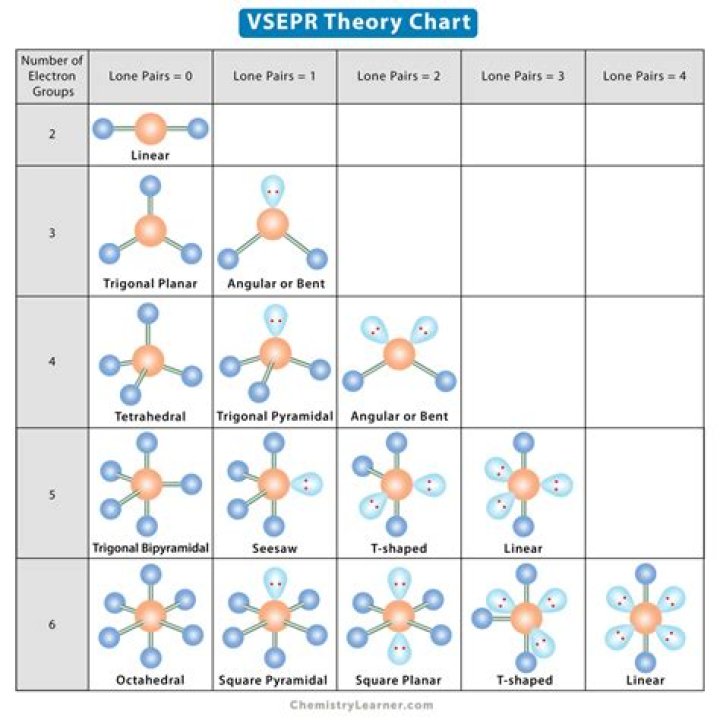
The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
What are the 4 Vsepr shapes?
The VSEPR model is useful for predicting and visualizing molecular structures. The structures are: linear, trigonal planar, angled, tetrahedral, trigonal pyramidal, trigonal bipyramidal, disphenoidal (seesaw), t-shaped, octahedral, square pyramidal, square planar, and pentagonal bipyramidal.
How do you identify a Vsepr shape?
- VSEPR Rules:
- Identify the central atom.
- Count its valence electrons.
- Add one electron for each bonding atom.
- Add or subtract electrons for charge (see Top Tip)
- Divide the total of these by 2 to find the total.
- number of electron pairs.
- Use this number to predict the shape.
What are the five ideal shapes of a molecule?
Of these shapes, the ones with no lone pairs are called the ideal shapes. The five ideal shapes are: linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. One important point to note about molecular shape is that all diatomic (compounds with two atoms) compounds are linear.What are the 6 types of molecule shapes?
- Linear. Ernest Wolfe. …
- Trigonal Planar. 4 total atoms: 1 central atom and 3 surrounding atoms. …
- Trigonal Bent. 3 total atoms: 1 central atom and 2 surrounding atoms. …
- Trigonal Pyramidal. …
- Tetrahedral. …
- Bent Tetrahedral.
What is a Vsepr formula?
AXE method. The “AXE method” of electron counting is commonly used when applying the VSEPR theory. The electron pairs around a central atom are represented by a formula AXnEm, where A represents the central atom and always has an implied subscript one. Each X represents a ligand (an atom bonded to A).
What is shape in chemistry?
The shape of a molecule affects its chemical and physical properties, including its color, reactivity, and biological activity. The bond angles between adjacent bonds may be used to describe a molecule’s overall shape.
What is the Vsepr shape of NH3?
Total:84 electron pairs:tetrahedral geometry for the four shape-determining electron pairsWhat shape is NF3?
The molecular geometry or shape of NF3 is a trigonal pyramid and its electron geometry is tetrahedral. NF3 lewis dot structure contains 1 lone pair and 3 bonded pairs.
What is the shape of h2o?H2O Water. View Live. Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
Article first time published onWhat shape is a tetrahedral?
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners.
What is AX3?
AX3 is a data logger. It features a state of the art MEMS 3-axis accelerometer and Flash based on-board memory. … The device is ideal for collecting longitudinal movement data.
How many Vsepr shapes are there?
Molecular Geometries. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
How do you memorize hybridization?
- If it’s 4, your atom is sp3.
- If it’s 3, your atom is sp2.
- If it’s 2, your atom is sp.
What shape is AX3E2?
T-Shaped (AX3E2) This is an example of a t-shaped molecule. These molecules consist of three atoms surrounding a central atom. The central atom has two lone pairs of electrons which make the angles close to 90 degrees.
What is the shape of BrF2?
Total Domains: 5 – Molecular Shape: Linear – Electron Geometry: Trigonal Bypyramid – Hybridization: sp^3d – Bond Angles: 90 and 120 – Example: BrF2- AX6.
What molecular shape is CHCl3?
CHCl3 would have an AX4 designation. This would make its shape tetrahedral. It’s just like CH4 but with Cl atoms replacing three of the hydrogens.
What shape is a atom?
We can say that an atom “has the shape of a sphere,” but it is not a sphere. This is because the outer layers of the atom, composed entirely of the electron orbitals, are essentially like a cloud, with different densities at different times, which we can’t precisely predict.
What is an atom vs molecule?
Atoms are single neutral particles. Molecules are neutral particles made of two or more atoms bonded together. An ion is a positively or negatively charged particle.
Is an atom a molecule?
A molecule is made up of atoms bonded together. So, while an atom is its own separate entity, a molecule is what you get when those atoms bond together. These might be the same elements, such as two oxygen atoms bonded together (O2), or it might be different atoms bonded together like water (H2O).
What is the difference between geometry and shape?
The key difference between shape and geometry of a molecule is that shape of a molecule is the structure of the molecule excluding the lone pair on the central atom whereas the geometry of a molecule describes the arrangement of lone pair and bond pair electrons around the central atom of the molecule.
What is shape in chemistry class 9?
(v) Shape: Solids have definite shape whereas liquids take the shape of the container in which they are placed and gases do not have any shape. … The particles of a gas have maximum kinetic energy due to free motion of gas particles in all directions. Solids have minimum kinetic energy due to least movement of particles.
Why do compounds have different shapes?
These shapes are decided by the arrangement of electrons around the central atom in the molecule. … The presence of electrons that are not involved in bonding around the central atom, so-called ‘lone pairs’, are closer to the central atom, and as such repel electron pairs around them more strongly.
Why is molecular shape important?
The location in three-dimensional space of the nucleus of each atom in a molecule defines the molecular shape or molecular geometry. Molecular shapes are important in determining macroscopic properties such as melting and boiling points, and in predicting the ways in which one molecule can react with another.
Is NH3 bent?
If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3). If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).
Why is VSEPR important?
Clearly it is very important to know the shape of a molecule if one is to understand its reactions. It is also desirable to have a simple method to predict the geometries of compounds. For main group compounds, the VSEPR method is such a predictive tool and unsurpassed as a handy predictive method.
Is VSEPR electron geometry?
The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. … We separate this into two categories, the electron-group geometry and the molecular geometry.
What are the rules of VSEPR theory?
VSEPR Rules. Draw the Lewis structure for the molecule or ion. Count the total number of regions of high electron density (bonding and unshared electron pairs) around the central atom. Double and triple bonds count as ONE REGION OF HIGH ELECTRON DENSITY.
What is the shape of cif3 and NF3?
So NF3 molecule has a trigonal pyramidal type of structure . the shape is pyramidal in case of NF3 but dont mix it with NCl3 because its shape is trigonal planar due to back bonding.
Is methane a planar?
The molecular geometry of the methane molecule is tetrahedral (see Figure 7). The H-C-H bond angles are 109.5°, which is larger than the 90° that they would be if the molecule was planar.
What is structure of nh3?
Ammonia is a covalent atom. The particle is shaped because of the overlap of orbitals of three hydrogen atoms and three Sp3 hybrid orbitals of nitrogen in the structure as the central atom. The fourth sp³ hybrid orbital is involved by a lone pair. Due to this the shape of ammonia is trigonal pyramidal.