What atoms are in CH3COOH
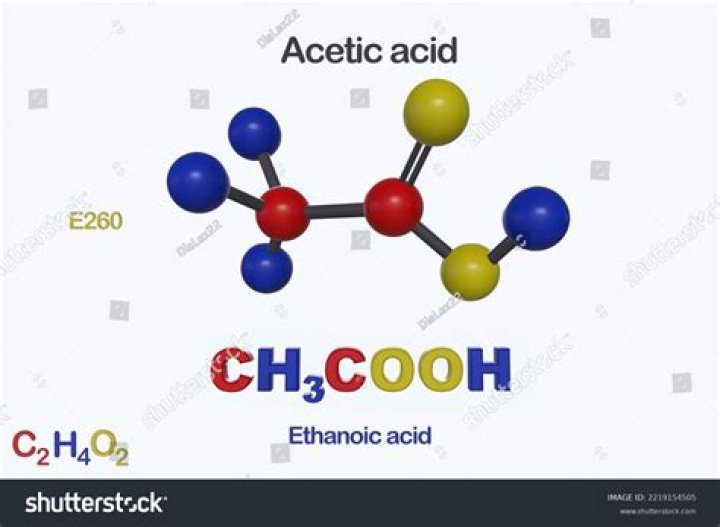
This formula indicates that a molecule of acetic acid (Figure 2.21) contains two carbon atoms, four hydrogen atoms, and two oxygen atoms.
How many atoms are contained in a mole of CH3COOH?
As the molecular formula of acetic acid is $ C{H_3}COOH $ , it is clear that it has two carbon atoms, two oxygen atoms, and four hydrogen atoms. Mole is used to express the amount of chemical substance. One mole consists of $ 6.02 \times {10^{23}} $ number of units, these units may be atoms, or molecules, or ions.
How many moles of atoms are in a mole of CH3COOH molecule?
In 1 mole of acetic acid, CH3COOH , 2 moles of oxygen atoms are present.
Is CH3COOH an atom or molecule?
In the CH3COOH molecule, three types of atoms are present – hydrogen, oxygen, and carbon.How many atoms are in 2 molecules CH3COOH?
CH3COOH is the Acetic Acid and it is organic compoud and weak acidic in nature. it has two carbon (C) atoms, four hydrogen (H) atoms and two oxygen (O) atoms so total 8 atoms.
How many atoms are in 1 mole of ch3coo c2 h5?
so 1 mole has 6×10^ -23 molecules of ethyl ethanoate.
How many extra elements are in CH3COOH?
CH3COOH is called Acetic acid, also known as ethanoic acid and methane carboxylic acid, is a colorless liquid that has a strong and distinct pungent and sour smell. It has two carbon (C) atoms, four hydrogen (H) atoms and two oxygen (O) atoms. Because it has a carbon in its chemical formula, it is an organic compound.
What is the intermolecular force of CH3COOH?
The intermolecular forces in CH₃CO₂H are an especially strong type of dipole-dipole force given its own special name — hydrogen bonding. Hydrogen bonds form when you have a negative O, N, or F atom in one molecule and a positive H atom attached to an O, N, or F atom in another molecule. Water has strong hydrogen bonds.What shape is CH3COOH?
Name of MoleculeAcetic acidMolecular Geometry1-tetrahedral, 2-trigonal planar, 3-bentHybridizationc1- sp3 , c2- sp2Molecular FormulaCH3COOHMolecular Weight60.052 g/mol
How many atoms of oxygen are in 300 molecules of CH3COOH?So, there are 600 atoms of oxygen in 300 molecules of acetic acid.
Article first time published onWhat is the number of oxygen atoms in 0.5 mol of CH3COOH?
There are two oxygen atoms in CH3COOH therefore two moles of oxygen atoms therefore two times Avogadro’s constant atoms. Equation of acetic acid:CH3COOH,(C2H4O2). so oxygen atoms are 2(TWO).
What is the percent by mass of hydrogen in CH3COOH?
The mass percent of hydrogen in the sample is 7.8%. The mass has been substituted correctly. The mass percent is reasonable and is correctly expressed to two significant digits. Calculate the mass percent of oxygen in acetic acid, CH3COOH(ℓ).
How many atoms are in a molecule of CH4?
Methane (US: , UK: ) is a chemical compound with the chemical formula CH4 (one atom of carbon and four atoms of hydrogen).
How many atoms does n2o?
Nitrous oxide—also called by various names such as hyponitrous acid anhydride, dinitrogen oxide, nitrogen monoxide, and factitious air—is the chemical compound represented as N2O; that is, composed of two atoms of nitrogen (N) and one atom of oxygen (O).
How many atoms are in 5nh3?
Answer: In one molecule of NH3 , there is 1 atom of N and 3 atoms of H , for a total of 4 atoms.
What is oxidation number of CH3COOH?
The chemical name of the given chemical compound is ethanoic acid. Ethanoic acid is commonly known as acetic acid. In this chemical compound, two carbon atoms are present, four hydrogen atoms are present, and two oxygen atoms are present. … Thus, the oxidation number of carbon of the carboxylic group is +3.
How many atoms are in one mole of caoh2?
∴1mole ofCa(OH)2 contains 5N atoms.
How many atoms are there in CaCO3?
Calcium carbonate has the formula CaCO3. This means that one molecule of Calcium Carbonate has 5 atoms in total – one calcium atom, one carbon atom, and 3 oxygen atoms.
Which has maximum number of atoms a 24g of C 12?
= 12.046 * 10^23 atoms. Assuming that there is no impurity present, the maximum number of atoms in that case would be 2×6.023×(10^22). Therefore the total number of atoms =24/12 moles=2moles =2×6.023×(10^22).
How many atoms are there in 0.25 mole of hydrogen?
So, 0.25 moles of hydrogen will have = 6.022 × 1023 × 0.25 = 1.50 × 1023 atoms of hydrogen.
What is the molecular mass of COOH?
As atomic mass of C= 12, O=16, and H=1 therefore 12+16+16+1= 45 hence, M.M is 45g/mol.
What is the percentage by number of atoms of carbon in Ethanoic acid CH3COOH?
The percent composition of carbon in acetic acid is 40.002% .
How many ions are in acetate?
PubChem CID175Molecular FormulaC2H3O2-Synonymsacetate Acetate Ion Acetic acid, ion(1-) 71-50-1 Acetate ions More…Molecular Weight59.04Parent CompoundCID 176 (Acetic acid)
How many central atoms are in glycine?
Glycine is an organic compound that contains 2 carbon atoms, 5 hydrogen atoms, 1 nitrogen atom, and 2 oxygen atoms. It is one of the 20 amino acids commonly found in animal proteins.
What is the geometry of an atom?
Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.
Is ch3cooh molecular or ionic?
Acetic acid is molecular but hydrogen is Ionic.
Is ch3cooh covalent?
Though acetic acid is covalent it dissociates into acetate ions and hydrogen ions when mixed with water. Acetate ions are responsible for conducting electricity.
What is the strongest intermolecular force in CH3COOH?
One of these (CH3COOH) has the ability to hydrogen-bond. It will probably have the strongest intermolecular forces. 2. CH3COOH is the only one of these molecules to have a dipole, and we already decided it hasthe strongest intermolecular forces.
Is CH3COOH a hydrogen bond?
Compoundacetic acidFormulaCH3CO2HMol. Wt.60Boiling Point118ºCMelting Point17ºC
Is CH3COOH a solid?
Acetic acid (CH3COOH) forms a molecular solid.
How many atoms are there in 300 molecules of water?
300 gr water is 300 gr/18 gr per mole = 16,67 moles of H2O so 16,67 moles of oxygen atoms and 1 mole has 6,022 14 × 10^23 atoms/particles.



