What is atomic radius trend
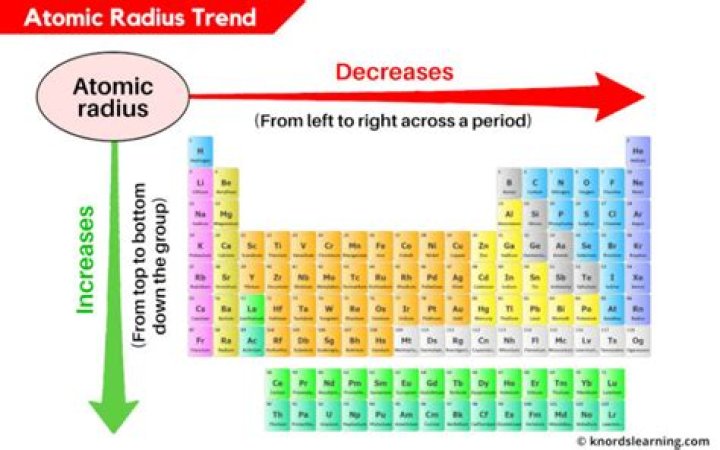
The atomic radius of atoms generally increases from top to bottom within a group. As the atomic number increases down a group, there is again an increase in the positive nuclear charge. … As the atomic number increases within a period, the atomic radius decreases.
What is the trend for atomic radius quizlet?
Group Trend: as you go down a column, atomic radius increases. Periodic Trend: as you go across a period, (L to R), atomic radius decreases. Group Trend: as you go down a column, ionization energy decreases.
What are the two trends in atomic radius?
There are two main atomic radius trends. One atomic radius trend occurs as you move left to right across the periodic table (moving within a period), and the other trend occurs when you move from the top of the periodic table down (moving within a group).
Why does the atomic radius trend make sense?
Atomic size gradually decreases from left to right across a period of elements. This is because, within a period or family of elements, all electrons are added to the same shell. However, at the same time, protons are being added to the nucleus, making it more positively charged.What is the greatest trend of atomic radius?
Atomic radii vary in a predictable way across the periodic table. As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. Thus, helium is the smallest element, and francium is the largest.
What is a periodic trend and group trend?
Periodic trends are specific patterns in the properties of chemical elements that are revealed in the periodic table of elements. … Periodic trends arise from the changes in the atomic structure of the chemical elements within their respective periods (horizontal rows) and groups (vertical columns) in the periodic table.
Why does atomic radius increase down a group quizlet?
Why does atomic radii increase as you move down a group? The number of energy levels increases as you move down a group as the number of electrons increases. Each subsequent energy level is farther from the nucleus than the previous one. Therefore, the atomic radius increases as the group and energy levels increase.
Why does reactivity decrease across a period?
Period – reactivity decreases as you go from left to right across a period. The farther to the left and down the periodic chart you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity. … Group – reactivity decreases as you go down the group.How do atomic radii vary in a group and period?
In general, the atomic radius decreases as we move from left to right in a period with an increase in the nuclear charge of the element. The atomic radius increases when we go down a group because of the addition of an extra shell.
What is atomic radius explain why it decreases across a period?The distance from the centre of nucleus to outermost shell of an atom is atomic radius. Atomic radius decreases across a period because the electron is added in the same shell. So attraction between the nucleus and valence shell increases due to which outermost shell is pulled in closer to the nucleus.
Article first time published onWhat is the trend in atomic radius across Period 3?
What is the trend in atomic radius of the elements across Period 3 and why does this occur? The atomic radius of the elements decreases from sodium to argon. This is because the number of protons increases (sodium has 11, argon has 18) so the nuclear charge increases.
What is the trend for ionic radius?
The size of an element’s ionic radius follows a predictable trend on the periodic table. As you move down a column or group, the ionic radius increases. This is because each row adds a new electron shell. Ionic radius decreases moving from left to right across a row or period.
How does the atomic radius increase?
An atom gets larger as the number of electronic shells increase; therefore the radius of atoms increases as you go down a certain group in the periodic table of elements. In general, the size of an atom will decrease as you move from left to the right of a certain period.
How does atomic size vary on the periodic table?
Explanation: Atomic size decreases across a Period from left to right as we face the Table, but INCREASES down a Group, a column of the Periodic Table. … And thus across the Period nucular charge predominates, and draws the valence electrons towards the nuclear core, with the result of a marked decrease in atomic radius.
What is the periodic trend observed in the variation of atomic radii down a group?
While going down the group the atomic radii goes on increasing, this is because new shells are added to the atoms of the elements as we go down from top to bottom in a group.
Why does atomic radius increase down Group 2?
Going down group 2: there are more filled shells between the nucleus and the outer electrons … … so the electrons in the higher energy levels are further from the nucleus … the atomic radius increases.
Why does an increase in atomic radius result in lower ionization energy?
Why do you think an increase in atomic radius would result in a lower ionization energy? Since the radius is larger, the electron is closer and does not have to travel as far to the positive nuclear charge, resulting in a lower ionization energy.
Why does the atomic radius shrink become smaller moving towards the left on the same row?
Explanation: As you move from left to right, the nucleus gains protons. This increases the positive charge of the nucleus and its attractive force on the electrons. … As a result, the valence electrons are held closer to the nucleus, and the atomic radius decreases.
How does the atomic radius change as you move down a group on the Periodic Table 2 points?
On the periodic table, atomic radius generally decreases as you move from left to right across a period (due to increasing nuclear charge) and increases as you move down a group (due to the increasing number of electron shells).
What is a periodic trend in chemistry?
Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties.
What is the relationship between atomic radius and ionization energy?
Moving left to right within a period or upward within a group, the first ionization energy generally increases. As the atomic radius decreases, it becomes harder to remove an electron that is closer to a more positively charged nucleus.
How do atomic radii vary in a group and why?
Atomic radius generally decreases from left to right across a period. … On the other hand, the atomic radius generally increases down a group. This is because down a group, the principal quantum number (n) increases which results in an increase of the distance between the nucleus and valence electrons.
What do you understand by atomic radii How does atomic radii vary a in a group B in a period explain Taking an example for each?
How do you explain the variation? Atomic radius of the elements generally decreases from left to the right in a period because on moving from left to right in a period the nuclear charge gradually increases by one unit and one electron is also added in the electron shell.
What is atomic radius How do the atomic radii of the elements change in a group?
In general, atomic radius reduces as one progresses through a period and increases as one progresses through a group. The number of energy levels (n) grows as one moves down a group, resulting in a greater distance between the nucleus and the outermost orbital. As a result, the atomic radius increases.
Does reactivity decrease down a group?
Period – reactivity increases as you go from the left to the right. Group – reactivity decreases as you go down the group.
Does reactivity increase down the group 1?
The reactivity of Group 1 elements increases as you go down the group because: … the attraction between the nucleus and outer electron gets weaker as you go down the group – so the electron is more easily lost.
What is the periodic trend for metal reactivity?
Metal reactivity decreases from left to right across periods and increases down groups. nonmetallic characteristics increases from left to right and decreases down groups. Nonmetal reactivity increases from left to right and decreases down groups.
What is meant by atomic radius explain?
The atomic radius of a chemical element is a measure of the size of its atoms, usually the mean or typical distance from the center of the nucleus to the boundary of the surrounding shells of electrons.
What is the trend as we go down Group 1?
Explaining the trend The reactivity of group 1 elements increases as you go down the group because: the atoms become larger. the outer electron becomes further from the nucleus. the force of attraction between the nucleus and the outer electron decreases.
How does atomic radii change across period 4?
The atomic radius of atoms generally decreases from left to right across a period. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. Within a period, protons are added to the nucleus as electrons are being added to the same principal energy level.
Which trends are correct across period 3 from Na to Cl?
First ionization energy decreases across period 3 from Na to Cl. Which increase across a period from left to right?



