What is Hemiketal and Ketal
As nouns the difference between ketal and hemiketal is that ketal is (chemistry) any acetal derived from a ketone while hemiketal is (chemistry) any hemiacetal derived from a ketone.
What is the difference between ketal and hemiketal?
As nouns the difference between ketal and hemiketal is that ketal is (chemistry) any acetal derived from a ketone while hemiketal is (chemistry) any hemiacetal derived from a ketone.
What is a hemiketal group?
A hemiacetal is a carbon connected to two oxygen atoms, where one oxygen is an alcohol (OH) and the other is an ether (OR). … This carbon chain could also only have hydrogen atoms attached, or it could have other functional groups such as alcohols.
What is meant by hemiacetal and ketal?
The Greek prefix hèmi means half, refers to the fact that a single alcohol has been added to the carbonyl group, in contrast to acetals or ketals, which are formed when a second alkoxy group has been added to the structure. Cyclic hemiacetals and hemiketals are sometimes called lactols.Is acetal and ketal the same?
The IUPAC originally deprecated the usage of the word ketal altogether, but has since reversed its decision. However, in contrast to historical usage, ketals are now a subset of acetals, a term that now encompasses both aldehyde- and ketone-derived structures.
What is ketal example?
In organic chemistry, a ketal is a functional group derived from a ketone by replacement of the carbonyl (C=O) group by two alkoxy groups. The IUPAC once declared the term “ketal” obsolete, but later accepted it as a subclass of acetals. Therefore, a ketal can also be defined as “an acetal derived from a ketone.”
Are Hemiacetal and Hemiketal same?
The key difference between Hemiacetal and Hemiketal is that hemiacetal is formed via the reaction between an alcohol and an aldehyde whereas a hemiketal is formed via the reaction between an alcohol and a ketone.
How do you distinguish or differentiate Ketal from acetal and Hemiketal from hemiacetal?
Acetal and hemiacetal are groups of atoms considered as functional groups. There is a slight difference between their chemical structures. The main difference between acetal and hemiacetal is that acetals contain two –OR groups whereas hemiacetals contain one –OR and one –OH group.How are Hemiacetal and Hemiketal formed?
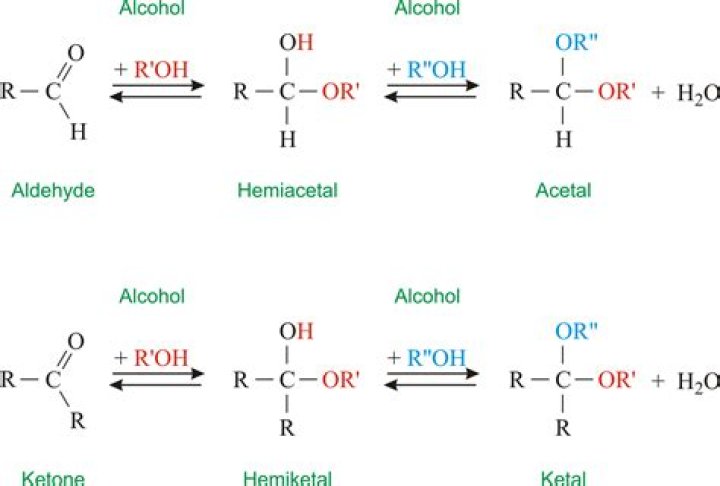
When an alcohol adds to an aldehyde, the result is called a hemiacetal; when an alcohol adds to a ketone the resulting product is a hemiketal. … A hydrate forms as the result of a water molecule adding to the carbonyl carbon of the aldehyde or ketone.
What is the structure of Ketal?An acetal is an organic molecule where two separate oxygen atoms are single bonded to a central carbon atom. Acetals have the general structure of R2C(OR’)2.
Article first time published onWhy are Hemiacetals important?
Hemiacetals and acetals are important functional groups because they appear in sugars. … First, an acid catalyst must be used because alcohol is a weak nucleophile; and second, the water produced with the acetal must be removed from the reaction by a process such as a molecular sieves or a Dean-Stark trap.
What is cyclization of monosaccharides?
Under normal biological conditions monsaccharides cyclize with only small amounts of the straight chain present in solution. When an aldose cyclizes, the hydroxyl group on the second to last carbon undergoes an intramolecular reaction with the carbonyl group of the aldehyde.
What is a ketal?
Definition of ketal : an acetal derived from a ketone.
What is the role of a strong acid in converting a Hemiketal to a ketal?
Hemiacetals and hemiketals can react with a second alcohol nucleophile to form an acetal or ketal. … The role of the acid catalyst is to protonate the OH group of the acetal, thus making it a good leaving group (water).
What is ketal reaction?
Ketal is a functional group which is derived from ketone by replacing the carbonyl group with two alkoxy groups. Ketal is prepared by the reaction of ketone with alcohols like methyl alcohol or ethyl alcohol under anhydrous condition. Ketone reacts with alcohol in presence of acid catalyst.
How is hemiketal formed?
They are formed when an alcohol oxygen atom adds to the carbonyl carbon of an aldehyde or a ketone. … When this reaction takes place with an aldehyde, the product is called a ‘hemiacetal’; and when this reaction takes place with a ketone, the product is referred to as a ‘hemiketal’.
What is Cyclic Ketal?
A cyclic ketal is a ketal in the molecule of which the ketal carbon and one or both oxygen atoms thereon are members of a ring. eg: see also cyclic acetal.
How do you make Ketal?
Ketals and acetals are formed by reaction of the carbonyl with alcohols such as methanol or ethanol under anhydrous conditions, in the presence of an acid catalyst. It is obvious that many alcohols can be used to generate acetals and ketals, but methanol and ethanol are probably the most common ones used.
Is Sucrose a hemiacetal or Hemiketal?
The alpha acetal is is really part of a double acetal, since the two monosaccharides are joined at the hemiacetal of glucose and the hemiketal of the fructose. There are no hemiacetals remaining in the sucrose and therefore sucrose is a non-reducing sugar.
What is a furanose ring?
The furanose ring is a cyclic hemiacetal of an aldopentose or a cyclic hemiketal of a ketohexose. A furanose ring structure consists of four carbon and one oxygen atom with the anomeric carbon to the right of the oxygen. … It is the opposite in an l-configuration furanose.
Is ketal a fructose?
Fructose, an important sugar isomeric with glucose, is a ketone, a ketohexose. … The ketal forms of fructose, however, are diethers, are not in equilibrium with open-chain ketone, do not show carbonyl reactions, and do not mutarotate. They are nonreducing derivatives of fructose.
Is Sucrose a ketal?
Sweet, sweet chemistry – Sucrose has both acetal and ketal functional groups.
What is the difference between Hemiacetals and acetals?
Acetals contain two –OR groups, one –R group and a –H atom. In hemiacetals, one of the –OR groups in acetals is replaced by a –OH group. This is the key difference between acetal and hemiacetal. Acetals and hemiacetals are two functional groups which are most commonly found in natural products.
How do you make cubane?
Synthesis. Allylic bromination with N-bromosuccinimide in carbon tetrachloride followed by addition of molecular bromine to the alkene gives a 2,3,4-tribromocyclopentanone. Treating this compound with diethylamine in diethyl ether causes elimination of two equivalents of hydrogen bromide to give the diene product.
How do you name Hemiacetals?
Hemiacetals are named substitutively as alkoxy-, aryloxy-, etc., derivatives of an appropriate hydroxy parent compound, such as an alcohol (see R-5.5. 1.1), and by functional class nomenclature in the same way as acetals (see R-5.6. 4.1) using the class name “hemiacetal”.
What are acetals used for?
Acetals are common carbonyl compound derivatives that are often used in Organic Synthesis as protecting groups for aldehydes and ketones, as well as in many other reactions.
Why are Hemiacetals unstable?
You can see why hemiacetals are unstable: they are essentially tetrahedral intermediates contain- ing a leaving group and, just as acid or base catalyses the formation of hemiacetals, acid or base also catalyses their decomposition back to starting aldehyde or ketone and alcohol.
What does cyclization mean?
: formation of one or more rings in a chemical compound.
What is Alpha D Glucopyranose?
Description. alpha-D-Glucose, also known as alpha-dextrose or alpha-D-GLC, belongs to the class of organic compounds known as hexoses. These are monosaccharides in which the sugar unit is a is a six-carbon containing moeity. alpha-D-Glucose is an extremely weak basic (essentially neutral) compound (based on its pKa).
How many Hexoses are there?
In the linear form, there are 16 aldohexoses and eight 2-ketohexoses, stereoisomers that differ in the spatial position of the hydroxyl groups. These species occur in pairs of optical isomers.
What type of reaction is tollen's test?
Tollens’ test, also known as silver-mirror test, is a qualitative laboratory test used to distinguish between an aldehyde and a ketone. It exploits the fact that aldehydes are readily oxidized (see oxidation), whereas ketones are not.