What is in a covalent bond
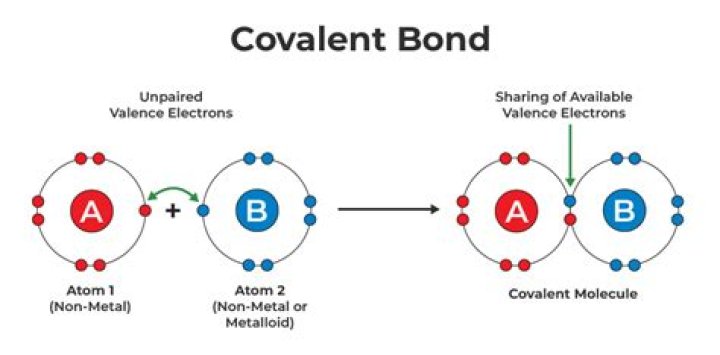
A covalent bond consists of the mutual sharing of one or more pairs of electrons between two atoms. These electrons are simultaneously attracted by the two atomic nuclei. A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions.
What is a covalent bond 8th grade?
A covalent bond is the force of attraction that holds together two atoms that share a pair of valence electrons. The shared electrons are attracted to the nuclei of both atoms. This forms a molecule consisting of two or more atoms. Covalent bonds form only between atoms of nonmetals.
What is covalent bond how it is formed?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.
What is a covalent bond GCSE definition?
A covalent bond forms when two non-metal atoms share a pair of electrons . The electrons involved are in the outer shells of the atoms. An atom that shares one or more of its electrons will complete its outer shell. Covalent bonds are strong – a lot of energy is needed to break them.What is covalent and ionic?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
What composes a covalent bond?
In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals. For example, in water (H2O) each hydrogen (H) and oxygen (O) share a pair of electrons to make a molecule of two hydrogen atoms single bonded to a single oxygen atom.
What is covalent bond and examples?
Examples of compounds that contain only covalent bonds are methane (CH4), carbon monoxide (CO), and iodine monobromide (IBr). Covalent bonding between hydrogen atoms: Since each hydrogen atom has one electron, they are able to fill their outermost shells by sharing a pair of electrons through a covalent bond.
How is the covalent bond formed answer in one sentence?
A covalent bond is formed by equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called shared pair or bonding pair.How do you represent covalent bonds?
Each covalent compound is represented by a molecular formula. The subscript is written only if the number is greater than 1., which gives the atomic symbol for each component element, in a prescribed order, accompanied by a subscript indicating the number of atoms of that element in the molecule.
What are covalent bonds quizlet?A bond formed between atoms where electrons are shared. … It happens between atoms of nonmetals. Molecule. The smallest particle of a substance that retains the chemical and physical properties of the substance and is composed of two or more atoms held together by chemical forces.
Article first time published onWhat is covalent bonding Class 10?
The chemical bond formed by the sharing of an electron pair between two atoms so that both the atoms get their octet complete is called covalent bond. The molecules formed as a result of sharing of electron between two atoms are called as covalent molecules.
What is a covalent bond save my exams?
Covalent bonds between atoms are very strong. When two or more atoms are chemically bonded together, they form ‘molecules’ Covalently bonded substances may consist of small molecules or giant molecules. Weak intermolecular forces exist between individual molecules.
What type of force is a covalent bond?
Covalent bonds: the binding electromagnetic force that arises between atoms when they closely share electrons in an approximately equal way.
What is difference between ionic bond and covalent bond?
Ionic bondsCovalent bondsA polar bond is formed by the attraction between oppositely-charged ions.Usually, an electron is more attracted to one atom than to another, forming a polar covalent bond.
What is the main difference between ionic and covalent bonding?
1. Ionic bonds result from transfer of electrons, whereas covalent bonds are formed by sharing. 2. Ionic bonds are electrostatic in nature, resulting from that attraction of positive and negative ions that result from the electron transfer process; charge separation between covalently bonded atoms is less extreme.
What is called covalent bond?
A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
What is covalent bond give two examples?
Difference Between Ionic and Covalent BondCovalent BondsIonic BondsCovalent Bonds are in Liquid or gaseous State at room temperatureAt room temperature, Ionic Bonds have Solid-state.Examples: Methane, Hydrochloric acidExample: Sodium chloride, Sulfuric Acid
What do you mean by covalent bond give example?
The chemcial bond formed by sharing of two valence electrons between the two atoms is called covalent bond. Example : Two hydrogen atoms share their electrons with each other to form H2 molecules . One covalent bond is formed between two hydrogen atoms by sharing of two electrons.
What elements are in a covalent bond?
- hydrogen.
- carbon.
- nitrogen.
- phosphorus.
- oxygen.
- sulfur.
- selenium.
What composed a covalent bond?
Covalent bonds consist of pairs of electrons shared by two atoms, and bind the atoms in a fixed orientation. … Whether two atoms can form a covalent bond depends upon their electronegativity i.e. the power of an atom in a molecule to attract electrons to itself.
How do you identify a covalent bond?
There is a couple different ways to determine if a bond is ionic or covalent. By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.
What is a covalent bond sentence?
Electrons in a covalent bond are essentially shared between the constituent atoms. Since the chlorine atom is much more electronegative than the hydrogen atom, the covalent bond between the two atoms is quite polar.
How does covalent bonding work?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.
What is a covalent bond Class 10?
A covalent bond is a chemical bond in which pairs of electrons are shared between two atoms. The covalent bond is also called a molecular bond.
What happens when a covalent bond forms quizlet?
A covalent bond forms when two or more valence electrons are attracted by the positively charged nuclei of two atoms and thus are shared between both atoms. … Molecules are stable when they achieve a noble gas configuration with an outer shell of 8 electrons.
What are the characteristics of covalent bonds quizlet?
- Formation of Bonds. Atoms have similar electronegativities so they will share electrons.
- Nonpolar. Share equally.
- Polar. Share unequally.
- Type of Atoms. Nonmetals.
- Melting Points. Low.
- Boiling Points. Low.
- Electrical Conductivity. Solid Phase- Nonconductors. …
- Solubility. Nonpolar- usually DO NOT dissolve in water.
What is covalent bond Class 11?
COVALENT BONDS. A covalent bond is a link between two atoms or two ions in which the electron pairs are shared between them. A covalent bond is also known as a molecular bond. Covalent bonds are formed between two non-metal atoms with identical or relatively close electronegativity values.
What is covalent bond by Vedantu?
In Chemistry, covalent bonds are formed between two atoms or ions in which the electron pairs are shared between them; they are also known as molecular bonds. The forces of attraction or repulsion between two atoms (when they share an electron pair or bonding pair) are called Covalent Bonding.
What is called ionic bond?
ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. … Sodium chloride exhibits ionic bonding.
What is metallic bonding a level?
Metallic bonding joins a bulk of metal atoms. Valence electrons are responsible for the metallic bond. Valence electrons can move from one atom to another creating a cloud of delocalized electrons. The positive metal ions are immersed in this cloud. … Metals can be drawn into thin wires, so we say that they are ductile.
What is a metallic bond Igcse?
Metallic bonds Metals form giant structures in which electrons in the outer shells of the metal atoms are free to move. The metallic bond is the force of attraction between these free-moving (delocalised) electrons and positive metal ions . … This is because the delocalised electrons can move throughout the metal.