What is Vsepr in chemistry
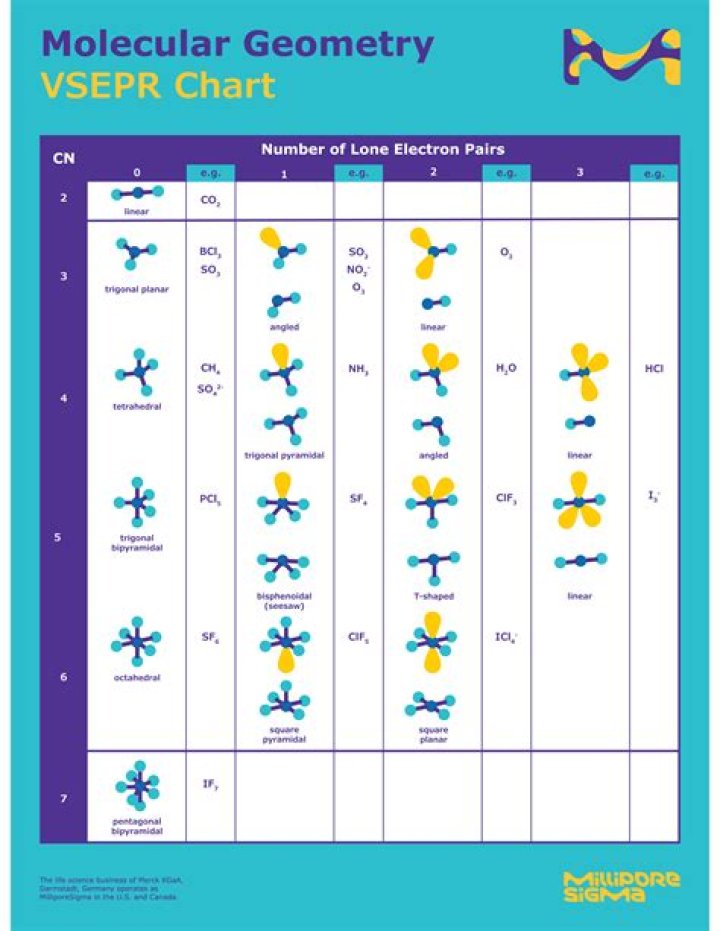
The valence shell electron pair repulsion (VSEPR) theory is a model used to predict 3-D molecular geometry based on the number of valence shell electron bond pairs among the atoms in a molecule or ion. … In other words, the electron pairs are as far apart as possible.
What is VSEPR theory simple?
The VSEPR theory is used to predict the shape of the molecules from the electron pairs that surround the central atoms of the molecule. The VSEPR theory is based on the assumption that the molecule will take a shape such that electronic repulsion in the valence shell of that atom is minimized. …
Why is VSEPR important?
Importance of VSEPR Models The VSEPR model predicts the 3-D shape of molecules and ions but is ineffective in providing any specific information regarding the bond length or the bond itself. … It can predict the shape of nearly all compounds that have a central atom, as long as the central atom is not a metal.
What is VSEPR theory with example?
VSEPR theory is used to predict the arrangement of electron pairs around central atoms in molecules, especially simple and symmetric molecules. … For example in the molecule methyl isocyanate (H3C-N=C=O), the two carbons and one nitrogen are central atoms, and the three hydrogens and one oxygen are terminal atoms.What are the steps for VSEPR?
- VSEPR Rules:
- Identify the central atom.
- Count its valence electrons.
- Add one electron for each bonding atom.
- Add or subtract electrons for charge (see Top Tip)
- Divide the total of these by 2 to find the total.
- number of electron pairs.
- Use this number to predict the shape.
Is VSEPR a successful scientific theory?
VSEPR theory is quite successful at predicting (or at least rationalizing) the overall shapes of molecules.
Why did the VSEPR theory fail?
The VSEPR model also fails to predict the structure of certain compounds because it does not take relative sizes of the substituents and stereochemically inactive lone pairs into account. Elements in the d-block have relatively high atomic masses and they tend to have stereochemically inactive electron pairs.
What shape is SF4?
Trigonal bipyramidal (sp3d) is the shape of SF4 with one equatorial position occupied by 1 lone pair. It has a see-saw shape as it contains four bond pairs and one lone pair.What is VSEPR number?
The valence shell electron pair repulsion (VSEPR) theory is a model used to predict 3-D molecular geometry based on the number of valence shell electron bond pairs among the atoms in a molecule or ion. This model assumes that electron pairs will arrange themselves to minimize repulsion effects from one another.
Which atom in a molecule is the focus of Vsepr theory?VSEPR stands for Valence Shell Electron Pair Repulsion Theory. We use VSEPR to predict the 3D shapes of the molecules made by the 2nd period elements. The main focus in this topic is going to be on the carbon (C), nitrogen (O), and oxygen (O).
Article first time published onWhat is the weakness of VSEPR in water?
The two main limitations of VSEPR theory include: Bond Angle Deviations: In the cases of methane and water, VSEPR predicts them both to be tetrahedral in shape. However, because of the bent shape of water, the bond angle is far less than that of methane and is something that VSEPR theory can’t account for numerically.
Under what circumstances does VSEPR work?
VSEPR works on the assumption that the shape adopted is that which minimises repulsions between the electron pairs in the molecule.
What are the 6 basic molecular shapes?
The 6 basic molecular shapes are linear, trigonal planar, angular (bent), tetrahedral, trigonal pyramidal, and trigonal bipyramidal.
Who discovered the VSEPR?
Ronald J. Gillespie (Fig. 1) of superacid and VSEPR fame is 85 years old this year.
Who invented VSEPR?
Gillespie did extensive work on expanding the idea of the Valence Shell Electron Pair Repulsion (VSEPR) model of Molecular Geometry, which he developed with Ronald Nyholm (and thus is also known as the Gillespie-Nyholm theory), and setting the rules for assigning numbers.
Who proposed VSEPR?
1.5: Structure and Bonding – Valence Shell Electron Pair Repulsion (VSEPR) Theory. The idea of a correlation between molecular geometry and the number of valence electrons was first presented in 1940 by Sidgwick and Powell; however, in 1957, Ronald Gillespie (Figure 1.5.
How many bonds can oxygen form?
Oxygen atoms form 2 covalent bonds because oxygen atoms have 6 valence electrons (2 lone pairs plus 2 unpaired electrons that are shared to achieve octet).
What is meant by bond pair?
A bond pair is a pair of electrons that are in a bond. A single bond is always composed of two electrons that are paired with each other. These two electrons together are called the bond pair. Bond pairs can be seen in covalent compounds and coordination compounds.
What is the shape of SF4 and xef2?
square planar and trigonal bipyramidal.
Is SF4 a seesaw?
An example of a seesaw shaped molecule is sulfur tetrafluoride, or SF4. Sulfur is the central atom, two fluorine atoms are on the equatorial plane, and two are on the axial plane.
Why is XeF4 square planar?
For XeF4 , there is a plane of symmetry so it is non-polar. Since there are 4 bonds to the central atom and 2 lone pairs, the geometry is square planar.
How will electron pairs behave according to VSEPR?
How will electrons pairs behave towards one another according to VSEPR? They will arrange themselves to be far apart from each other. … The repulsion between different types of electron pairs is always the same in magnitude.
Does VSEPR apply to ionic compounds?
No, but any polyatomic ions in them have a molecular geometry.
What is VSEPR theory class 11?
Hint: VSEPR theory is basically used to predict the geometry of molecules this theory assumes that each atom in a molecule will achieve a geometry that minimizes the repulsion between electrons in the valence shell of that atom. Complete answer: … VSEPR is the abbreviation of Valence Shell Electron Pair Repulsion theory.
What are the limitations of molecular orbital theory?
MO theory says that the electrons are delocalized. That means that they are spread out over the entire molecule. The main drawback to our discussion of MO theory is that we are limited to talking about diatomic molecules (molecules that have only two atoms bonded together), or the theory gets very complex.
Is Vsepr electron geometry?
The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. … We separate this into two categories, the electron-group geometry and the molecular geometry.
What are the 5 shapes of molecules?
Molecular Geometries. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
What is the molecular structure of o3?
The molecular geometry of o3 is trigonal planner.It is due to the presence of lone pair on the central metal atom which repels the electrons in the two bonds, causing the atom to adopt a bent molecular geometry..
Why is molecular geometry important?
It is important to be able to predict and understand the molecular structure of a molecule because many of the properties of a substance are determined by its geometry. … Molecular geometry may also be used to predict biological activity, to design drugs or decipher the function of a molecule.



