What type of bond does N2
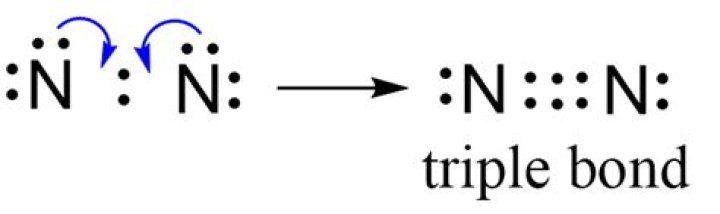
The N2 Lewis structure has a triple bond between two nitrogen atoms. According to the octet rule, nitrogen atoms need to bond three times. The N2 molecule is diatomic, meaning that two atoms of the same element are connected in a pair.
Is N2 covalent bond?
Nitrogen has 5 valence electrons therefore needs three more electrons to complete its octet configuration. Mutual sharing of three pair of electrons gives triple covalent bond.
How many covalent bonds are in N2?
Nitrogen typically forms 3 covalent bonds, including in N2 . This is because it has atomic number 7, so its electron configuration is 1s22s22p3 , giving it 5 valence shell electrons.
What is N2 ionic or covalent?
N2 forms 3 covalent bonds, it shares 3 electrons with each other of the nitrogens.Is N2 a molecule or compound?
Nitrogen gas (N2) is a molecule because the bond between the nitrogen atoms is a molecular bond. Water (H2O) is a molecular compound because it is a substance made from more than one kind of element that is held together with molecular bonds.
Is NO2 an ionic compound?
NO2 is covalent bond. because the electronegativity difference between the nitrogen and oxygen in NO2 is not enough to cause an ionic interaction,so it can’t be ionic bond.
What type of bond is N2 polar or nonpolar?
It is non-polar, because it is made up of two identical nitrogen atoms, both of which have the same electronegativity. Therefore none of the nitrogen atoms pull the electrons at a greater strength towards its nucleus than the other, and so the bond is not polar.
What is N2 in chemistry?
Molecular nitrogen (N2) is a very common chemical compound in which two nitrogen atoms are tightly bound together. Molecular nitrogen is a colorless, odorless, tasteless, and inert gas at normal temperatures and pressures. Four representations chemists use for nitrogen molecules.What type of bond is CuCl2?
Chlorine has a high electro negativity of 3.0. Copper like most metals has a low electro negativity, So the bonding is ionic making the compound an ionic salt. Ernest Z. CuCl2 is a covalent compound.
What type of solid is N2?Small symmetrical molecules (nonpolar molecules), such as H2, N2, O2, and F2, have weak attractive forces and form molecular solids with very low melting points (below −200 °C).
Article first time published onAre O2 and N2 polar molecules?
Generally, bonds between two of the same atoms, such as nitrogen (N2) or oxygen (O2) have an even distribution of electrons, making the atoms non-polar. Other molecules that use more than two of the same atom, such as ozone (O3), are also non-polar.
What is the electronegativity of N2?
The first scale of electronegativity was developed by Linus Pauling and on his scale nitrogen has a value of 3.04 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine).
What kind of bond type is NO2?
The nitrogen dioxide is a covalent compound where one nitrogen is the central atom which is bonded to two oxygen atoms, where one oxygen atom is bonded by a single bond and other oxygen atom by a double bond.
Is NO2 an element or compound?
It is a molecular compound as it is composed of two different elements bonded to one another.
Is AgCl a covalent compound?
Hence AgCl is ionic in nature and referred as an ionic compound. … A metal and a non-metal atom forms an ionic bond whereas atoms with less electronegativity will form covalent bonds. The sharing of electrons between the atoms will result in a covalent bond.
Is cucl2 covalent bond?
The chemical formula shows the compound copper(II) chloride. The compound can be decided to be ionic or covalent based on the difference in electronegativity. The electronegativity of copper is 1.9. … Therefore, each copper atom loses two electrons for two chlorine atoms.
Is p4o10 ionic or covalent?
Phosphorus pentoxide is a covalent compound that is composed of 4 phosphorus (P) atoms and 10 oxygen (O) atoms.
Is N2 heterogeneous or homogeneous?
ABsalt waterhomogeneous mixturedinner saladheterogeneous mixturecarbon dioxidecompoundnitrogenelement
Is N2 dinitrogen or nitrogen?
Given the great reactivity of atomic nitrogen, elemental nitrogen usually occurs as molecular N2, dinitrogen.
How is N2 formed?
The main reaction path for the N2 formation is the attack of N2O to carbon free site, forming N2 with no NtN splitting and leaving oxygen on the carbon surface. N2O is a key substance that affects the depletion of the ozone layer as well as global warming as a result of greenhouse effect.
Is SCl2 polar or nonpolar?
SCl2 is a polar molecule. There is a mild dipole because of the difference in electronegativity of Cl – 3.0 and S – 2.58. The molecule is bent (103^ bond angle) with the two chlorines, each with a partial negatives because of electron withdrawal, on one side of the molecule.
What type of bond would N2 have based on its electronegativity difference?
Explanation: Because the electronegativity difference is zero. When the electronegativity is between 0 – 0.5 the bond would be considered as a covalent bond. Well, from what does polarity result?



