Where are metalloids found
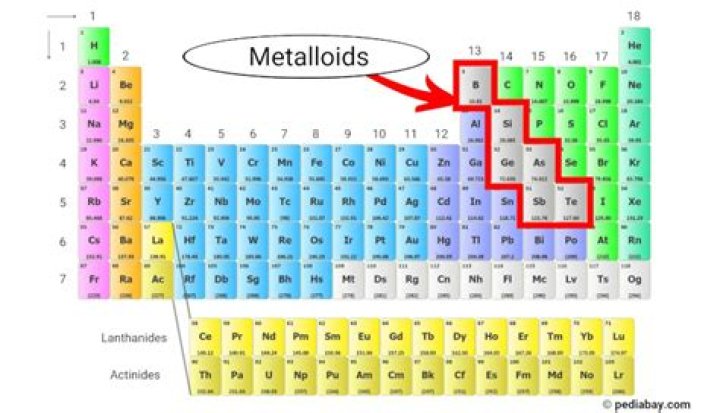
The metalloids are a group of elements in the periodic table. They are located to the right of the post-transition metals and to the left of the non-metals.
What is the most common metalloid and where is it found?
Silicon, immediately below carbon in group 4A, is by far the most abundant metalloid, being present in over 27% of the Earth’s crust. Silicon forms strong bonds with oxygen. Over 60% of silicon is present as feldspars and aluminosilicates, aluminum able to replace silicon because of its similar atomic radius.
What are metalloids and where are they located in the periodic table?
A metalloid is an element that has properties that are intermediate between those of metals and nonmetals. Metalloids can also be called semimetals. On the periodic table, the elements colored yellow, which generally border the stair-step line, are considered to be metalloids.
Where are metalloids found on the periodic table quizlet?
Metalloids are found in Groups 13 through 17 on the periodic table.Where in industry are metalloids mostly used?
Some of the metalloids (silicon and germanium) are semiconductors making them useful in chips for the semiconductor industry. Other metalloids are often used as dopants for semiconductor manufacturing.
What are metalloids give an example?
A metalloid is a chemical element that exhibits some properties of metals and some of nonmetals. … Boron, silicon, germanium, arsenic, antimony, tellurium, and polonium are metalloids.
Which of the following elements are metalloids?
Germanium is a metalloid.
Which group on the periodic table contain metalloids?
Groups 13–16 of the periodic table contain one or more metalloids, in addition to metals, nonmetals, or both. Group 13 is called the boron group, and boron is the only metalloid in this group. The other group 13 elements are metals. Group 14 is called the carbon group.What are metalloids on the periodic table quizlet?
A metalloid is an element that has physical and chemical properties of both metals and nonmetals. The elements boron, silicon, germanium, arsenic, antimony, tellurium, polonium, and astatine are metalloids.
What are metalloids quizlet?The metalloids are a unique group of elements that share properties of both metals and nonmetals. They’re also called the semimetals because of the shared properties of these elements along the dividing line between metals and nonmetals.
Article first time published onWhy are the metalloids located where they are on the periodic table?
The elements that are pea-green in colour, boron, silicon, germanium, arsenic, etc. are metalloids. And these have intermediate properties with respect to metals and non-metals.
Where are the non metalloids located on the periodic table?
1) METALS- located on the left side of the periodic table. 2) NONMETALS- located on the right side of the periodic table. 3) METALLOIDS- located on a “staircase” between metals and nonmetals.
How does the physical location of metalloids in the periodic table reflect the properties of metalloids?
Metalloids are all solid at room temperature. They can form alloys with other metals. … The physical properties of metalloids tend to be metallic, but their chemical properties tend to be non-metallic. The oxidation number of an element in this group can range from +5 to -2, depending on the group in which it is located.
What are examples of metalloids in everyday life?
- Computer components: Silicon is a semiconductor, making it ideal for computer chips and other electronic components. …
- Rubber: Silicon forms a polymer when mixed with oxygen. …
- Automotive engines: Alloys of aluminum and silicon are a common choice for building engine parts, including the block and cylinder head.
What is the most common metalloid on our planet?
Silicon is the “big sister” to carbon, and shares many of the same chemical properties. For this reason, silicon is known as a chemical analogue to carbon. But unlike carbon, silicon a metalloid — in fact, it’s the most common metalloid on earth.
Why are metalloids used in the electronics industry?
Metalloids have a key role in modern industry. Silicon and germanium metalloids are used as a semiconductors because they allow a controlled flow of electricity through them which is a useful property. Semiconductors have become very essential aspect of modern life due to their importance in electronics.
Which of the following is a metalloid *?
The elements most often regarded as metalloids are boron, silicon, germanium, arsenic, antimony and tellurium. Wikipedias generally categorises these six as metalloids, with the addition of astatine.
Why boron is a metalloid?
Boron (B) is a metalloid bcz it has characteristics of both metals & non-metal. Boron acts as non-metal when it reacts with highly electro-positive metals as Na, K etc. & B acts as metal when it reacts with F (to produce BF3).
Which of the following elements is not metalloid?
Carbon is not a metalloid and is a non-metal. Metalloids are those elements possessing properties between non-metals and metals. These are also known as semi-metals. The elements which are metalloids, in the periodic table are boron, silicon, arsenic, antimony, polonium, and tellurium.
What are metalloids Class 8 examples?
- Boron (B)
- Silicon (Si)
- Germanium (Ge)
- Arsenic (As)
- Antimony (Sb)
- Tellurium (Te)
- Polonium (Po)
Is an example of metalloid *?
Silicon, mercury, Diamond, Sulphur, Iodine, Germanium, Sodium, carbon, magnesium, Copper, Boron, Helium.
What is metalloid Class 9?
Metalloids are the elements which are having a combination of some properties of metals along with some properties of nonmetals. Their properties are intermediate between the properties of metals and nonmetals, they are also called semi-metals. Example: – Boron, Silicon, Germanium, etc.
Which element in period three on the periodic table is a metalloid?
Silicon. Silicon (symbol Si) is a group 14 metalloid. It is less reactive than its chemical analog carbon, the nonmetal directly above it in the periodic table, but more reactive than germanium, the metalloid directly below it in the table.
What properties do metalloids share with metals?
Physical Properties of Metalloids They fall between metals and nonmetals in their ability to conduct heat, and if they can conduct electricity, they usually can do so only at higher temperatures. Metalloids that can conduct electricity at higher temperatures are called semiconductors.
What are the general properties of a metalloids quizlet?
A metalloid is a chemical element with properties that are in between metals and nonmetals. The ability or power to conduct or transmit heat, electricity, or sound. The state of being malleable, or capable of being shaped, as by hammering or pressing without breaking. The state or quality of not being shiny.
What element is the only metalloid in Group 17?
Tellurium is the only metalloid in this group, which also contains three nonmetals and one metal.
Which group contains metals metalloids and nonmetals?
Group 12 consists of metals (Zinc, Cadmium, mercury). So, we get to know that group 14 consists of metals, nonmetals and metalloids.
Why is carbon a metalloid?
Carbon is a non-metal. It belongs to the fourteenth group or IV A group in the modern periodical table. The elements of this group have four electrons in the valence shell. … To get the octet in its outer shell it has to gain four more electrons to form C4-.
What property makes metalloids important?
Metalloids are typically semi-conductors, which means that they both insulate and conduct electricity. This semi-conducting property makes Metalloids very useful as a computer chip material.
Why are metalloids located between metals and nonmetals?
The metalloids, or semimetals, have properties that are somewhat of a cross between metals and nonmetals. Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry.
Where on the periodic table can you find the metalloids semimetals?
The metalloids or semimetals are located along the line between the metals and nonmetals in the periodic table.



