Who did Millikan work with
Robert A. MillikanInstitutionsUniversity of Chicago California Institute of TechnologyThesisOn the polarization of light emitted from the surfaces of incandescent solids and liquids. (1895)Doctoral advisorOgden Nicholas RoodOther academic advisorsMihajlo Pupin Albert A. Michelson Walther Nernst
What other scientists did Robert Millikan work with?
His doctoral research concerned the polarization of incandescent light. In 1895, Millikan moved to Germany and studied for a year with Max Planck and Walther Nernst at the Universities of Gottingen and Berlin, returning to the U.S. in 1896 to join A. A. Michelson at the University of Chicago.
What did Robert A Millikan contribute?
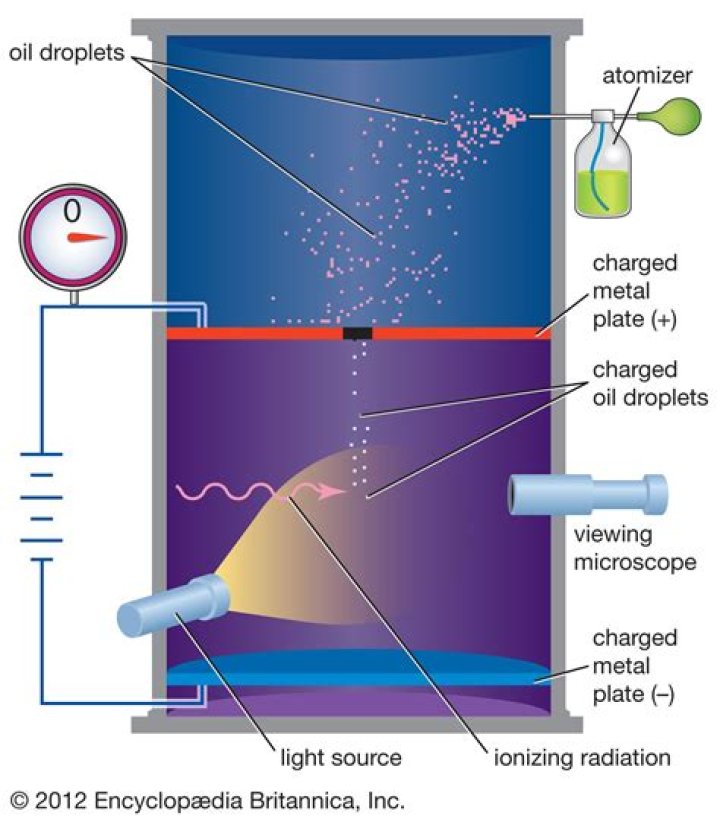
Robert Millikan was a physicist who discovered the elementary charge of an electron using the oil-drop experiment.
Who worked on the elementary charge using the photoelectric effect?
Robert Millikan, in full Robert Andrews Millikan, (born March 22, 1868, Morrison, Illinois, U.S.—died December 19, 1953, San Marino, California), American physicist honoured with the Nobel Prize for Physics in 1923 for his study of the elementary electronic charge and the photoelectric effect.How did Millikan prove Einstein's theory?
While it had been known for a long time that light falling on metal surfaces may eject electrons from them (the photoelectric effect), Millikan was the first to determine with great accuracy that the maximum kinetic energy of the ejected electrons obey the equation Einstein had proposed in 1905: namely, 1/2mv2=hf-P, …
What did James Chadwick discover?
In 1932, Chadwick made a fundamental discovery in the domain of nuclear science: he proved the existence of neutrons – elementary particles devoid of any electrical charge.
What is James Chadwick atomic theory?
Chadwick is best known for his discovery of the neutron in 1932. A neutron is a particle with no electric charge that, along with positively charged protons, makes up an atom’s nucleus. … In this way, Chadwick’s findings were pivotal to the discovery of nuclear fission, and ultimately the development of the atomic bomb.
How did Marie Curie contribute to the atomic theory?
Radioactivity, Polonium and Radium Curie conducted her own experiments on uranium rays and discovered that they remained constant, no matter the condition or form of the uranium. The rays, she theorized, came from the element’s atomic structure. This revolutionary idea created the field of atomic physics.When did Robert Millikan do his work?
During the 1890s the theory that electricity was conveyed by a miniscule unit, the electron, gained acceptance. In 1910 Robert Millikan succeeded in precisely determining the magnitude of the electron’s charge.
When did Erwin Schrodinger make his discovery?His great discovery, Schrödinger’s wave equation, was made at the end of this epoch-during the first half of 1926. It came as a result of his dissatisfaction with the quantum condition in Bohr’s orbit theory and his belief that atomic spectra should really be determined by some kind of eigenvalue problem.
Article first time published onWhat did Robert Millikan do with the atomic theory?
Millikan discovered that there is a fundamental electric charge—the charge of an electron. Rutherford’s gold foil experiment showed that atoms have a small, dense, positively charged nucleus; the positively charged particles within the nucleus are called protons.
What is Erwin Schrodinger atomic theory?
Erwin Schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. … Electrons have an intrinsic property called spin, and an electron can have one of two possible spin values: spin-up or spin-down. Any two electrons occupying the same orbital must have opposite spins.
What did Erwin Schrodinger discover?
Assuming that matter (e.g., electrons) could be regarded as both particles and waves, in 1926 Erwin Schrödinger formulated a wave equation that accurately calculated the energy levels of electrons in atoms.
Did Millikan discover the mass of an electron?
Excellent question regarding Millikan’s oil drop experiment! Robert Millikan was able to determine the mass of an electron by using charged oil drops. Electrons have such a small mass, they were believed to be massless. Millikan’s experiment determined that the electrons did, indeed, have mass.
Who did James Chadwick work with?
Chadwick was educated at the University of Manchester, where he worked under Ernest Rutherford and earned a master’s degree in 1913. He then studied under Hans Geiger at the Technische Hochschule, Berlin.
Did James Chadwick win Nobel Prize?
The Nobel Prize in Physics 1935 was awarded to James Chadwick “for the discovery of the neutron.”
What did JJ Thompson discover?
Thomson, in full Sir Joseph John Thomson, (born December 18, 1856, Cheetham Hill, near Manchester, England—died August 30, 1940, Cambridge, Cambridgeshire), English physicist who helped revolutionize the knowledge of atomic structure by his discovery of the electron (1897).
Who disagreed with James Chadwick?
He clashed with fellow ACAE member Patrick Blackett, who disagreed with Chadwick’s conviction that Britain needed to acquire its own nuclear weapons; but it was Chadwick’s position that was ultimately adopted. He returned to Britain in 1946, to find a country still beset by wartime rationing and shortages.
What did Werner Heisenberg discover?
Werner Heisenberg discovered the uncertainty principle, which states that the position and the momentum of an object cannot both be known exactly.
How did James Chadwick prove the existence of neutrons?
To prove that the particle was indeed the neutron, Chadwick measured its mass. He could not weigh it directly. Instead he measured everything else in the collision and used that information to calculate the mass. For his mass measurement, Chadwick bombarded boron with alpha particles.
What did Henry Moseley discover?
In 1913, while working at the University of Manchester, he observed and measured the X-ray spectra of various chemical elements using diffraction in crystals. Through this, he discovered a systematic relation between wave- length and atomic number. This discovery is now known as Moseley’s Law.
What did Robert Millikan discover about the electron quizlet?
What did Robert Millikan discover about the electron? The charge and the mass of the electron. The charge on each drop was always some multiple of a single very small value, which he proposed to be the fundamental unit of charge carried by each e-.
What did Marie Curie work as?
Marie Curie was a physicist, chemist and pioneer in the study of radiation. She discovered the elements polonium and radium with her husband, Piere.
Did Marie Curie discover xrays?
X-Rays. Marie Curie not only made huge contributions to the fields of physics and chemistry, but also to the world of medicine. … [2] Curie worked on the X-ray machine discovered by German scientist Wilhelm Roentgen in 1895. She used her newly discovered element, radium, to be the gamma ray source on x-ray machines.
What did Marie Curie do?
Indefatigable despite a career of physically demanding and ultimately fatal work, she discovered polonium and radium, championed the use of radiation in medicine and fundamentally changed our understanding of radioactivity. Curie was born Marya Skłodowska in 1867 in Warsaw.
Is Olivia Newton John related to Max Born?
Max BornRelativesOlivia Newton-John (granddaughter)
Did Erwin Schrödinger have siblings?
Erwin Schrödinger was born on August 12, 1887 in Vienna, Austria to parents Georgine Emilia Brenda and Rudolf Schrödinger. Schrödinger did not have any siblings and was homeschooled by private tutors until the age of 11.
Who came first Rutherford or Millikan?
Two thousand years later, Dalton proved Democritus was correct. Shortly after that, electrons were discovered by Thomson, the nucleus was discovered by Rutherford and the charge of an electron was measured by Millikan.
Who discovered neutron?
By 1920, physicists knew that most of the mass of the atom was located in a nucleus at its center, and that this central core contained protons. In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron.
What was JJ Thomson atomic theory?
Summary. J.J. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.”
What was Ernest Rutherford atomic theory?
Ernest Rutherford found that the atom is mostly empty space, with nearly all of its mass concentrated in a tiny central nucleus. The nucleus is positively charged and surrounded at a great distance by the negatively charged electrons.



