Why is anthracene a diene
Anthracene, however, is an unusually unreactive diene. This is due to both steric effects, but more importantly because the “diene” is really part of an aromatic ring system and is thus stabilized. This stabilization in the reactant reduces the reactivity (stability/reactivity principle).
Is anthracene a conjugated diene?
Being an easily available conjugated π-electron-rich carbocyclic system, anthracene (1a) has been widely exploited as a classic diene in Diels–Alder reactions wherein its chemical reactivity and transformational effectiveness are subsidized by the partial loss of aromaticity.
What is a good diene?
Good friends are loyal and accept you for who you are during the good and bad times. Good friends are also honest — honest enough to tell you when you’re not being a good friend yourself. … Along with good friends who are present, loyal, and honest, most people want friends who are trustworthy.
Does anthracene undergo Diels reaction?
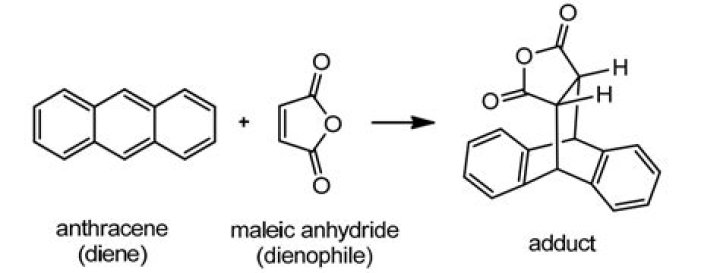
Anthracene undergoes a Diels-Alder reaction with maleic anhydride to give a cycloadduct with the formula C18H12O3. … The Diels-Alder reaction is the conjugate addition of an alkene to a diene. The alkene that adds to the diene is called dienophile.Is anthracene a diene or dienophile?
Anthracene acts as the diene and maleic anhydride functions as the dienophile. Xylene (dimethylbenzene) is used as a high boiling temperature solvent so that the reaction will proceed quickly .
Is anthracene polar or nonpolar?
Naphthalene and anthracene are non-polar, so soluble in non-polar solvents like benzene.
Is anthracene a cyclic compound?
Anthracene is anthracene, also called paranaphthalene or green oil, a solid polycyclic aromatic hydrocarbon (PAH) consisting of three benzene rings derived from coal-tar, is the simplest tricyclic aromatic hydrocarbon. It is an ortho-fused polycyclic arene and a member of acenes. …
What is the use of anthracene?
Anthracene is a solid polycyclic aromatic hydrocarbon (PAH) of formula C14H10, consisting of three fused benzene rings. It is a component of coal tar. Anthracene is used in the production of the red dye alizarin and other dyes.Why is anthracene considered to be an aromatic compound?
Anthracene is solid in state and consists of polycyclic aromatic hydrocarbon. It has three fused benzene rings derived from coal tar. Complete step by step answer: … For a compound considered aromatic, it follows Huckel’s rule and overlapping p orbitals in order to be aromatic.
How is anthracene synthesized?Anthracene can be synthesized by the Elbs reaction, in which o-tolyl phenyl ketone is dehydrated at 400–450 ºC. … In commerce, anthracene is mainly used as a starting material for the manufacture of 9,10-anthraquinone, which in turn is used to make colorants such as the red dye alizarin.
Article first time published onWhat is Dienes in chemistry?
In organic chemistry a diene (/ˈdaɪ. iːn/ DY-een) (diolefin (/daɪˈoʊləfɪn/ dy-OH-lə-fin) or alkadiene) is a covalent compound that contains two double bonds, usually among carbon atoms. They thus contain two alkene units, with the standard prefix di of systematic nomenclature.
What is the product of maleic anhydride and anthracene?
In this experiment you will learn how to use the Diels-Alder reaction to form a bridged polycyclic anhydride. Two solids – anthracene and maleic anhydride – will be dissolved in xylene and refluxed. The product 9,10-dihydroanthracene-9,10-α,β-succinic anhydride will be recovered and isolated by vacuum filtration.
What is the formula of anthracene?
Anthracene is a solid polycyclic aromatic hydrocarbon (PAH) of formula C14H10, consisting of three fused benzene rings. It is a component of coal tar.
What are Dienes and Dienophiles?
As nouns the difference between diene and dienophile is that diene is (organic chemistry) an organic compound, especially a hydrocarbon, containing two double bonds while dienophile is (organic chemistry) a compound that readily reacts with a diene; especially an alkene in the diels-alder reaction.
Is anthracene a sublimate?
Complete answer: -In the option (C) Iodine, camphor, ammonium chloride, anthracene and naphthalene are given and all these substance are sublime substance as they are present in solid state and possess sublimation process by directly converts into the gaseous state without changing into liquid state.
Is anthracene a tricyclic?
anthracene, a tricyclic aromatic hydrocarbon found in coal tar and used as a starting material for the manufacture of dyestuffs and in scintillation counters.
Is anthracene neutral?
We also know that Anthracene is a solid polycyclic aromatic hydrocarbon compound. As Anthracene is present naturally without any reaction with coal tar then it is neutral in nature.
Is anthracene soluble in dichloromethane?
Anthracene is insoluble in water but is quite soluble in carbon disulfide and somewhat soluble in ethanol, methanol, benzene, chloroform, and other organic solvents.
Is anthracene a planar molecule?
Since ALL of the carbons are this way, we can conclude that anthracene is a planar compound.
What is the PKA of anthracene?
Evaluation of pKH2O = 7.4 for anthracene is described in the previous paper. Combining this value with pKR = −6.0 gives pKa = −13.4.
Is anthracene is aromatic or not?
Anthracene is a solid polycyclic aromatic hydrocarbon (PAH) consisting of three fused benzene rings. … Anthracene is used in the production of the red dye alizarin and other dyes. Anthracene is colorless but exhibits a blue (λmax = 400–450 nm) fluorescence under ultraviolet (UV) light.
What is the aromaticity of anthracene?
Anthracene is aromatic. It has planar cyclic conjugated system of 14 pi electrons. A cyclic planar compounds with a conjugated system of 4n + 2 pi electrons is aromatic. Note: 4n+2=4(3)+2=14.
What are the chemical properties of anthracene?
Chemical Properties ANTHRACENE is a colorless solid; melting point 218 °C, blue fluorescence when pure; insoluble in water, slightly soluble in alcohol or ether, soluble in hot benzene, slightly soluble in cold benzene; transformed by sunlight into para -anthracene (C14H10)2.
How is anthracene used in everyday life?
Anthracene’s uses include use as a preservative in wood and lumber, and use as an insecticide for crops. Anthraquinone, derived directly from anthracene, finds use as a precursor to a lot of dyes the fabrics and textiles industries rely on.
Why are naphthalene and anthracene regarded as aromatic compounds?
Naphthalene is a bicyclic aromatic hydrocarbon having a resonance stabilization energy per ring slightly less than that of benzene (36 kcal/mole). … The resonance stabilization energy for each compound is again less than three times that of benzene, with that for anthracene being less than that of phenanthrene.
How is anthracene purified?
Anthracene is purified by sublimation. In sublimation, a solid is converted directly into gaseous state on heating without passing through liquid phase.
What is the isomer of anthracene?
Phenanthrene is an LMW-PAH, which is isomeric with anthracene in its chemical structure and appears as a white/yellow crystalline substance.
How many resonating structures are there for anthracene?
Anthracene will undergo delocalisation to give four resonance structures.
Why does anthracene undergo addition reaction easily?
These two positions are the most susceptible for the addition reactions, because when any atom attaches here replacing the hydrogen atom in the ring the resonance structures are more than the scenario where that same atom attaches any other positions. Thus making the product more stable.
What are Dienes with examples?
Dienes are compounds whose molecules contain two carbon-carbon double bonds separated by a single bond. The most important diene polymers—polybutadiene, polychloroprene, and polyisoprene—are elastomers that are made into vulcanized rubber products.
What are Dienes classify them?
Dienes can divided into three classes: Unconjugated dienes have the double bonds separated by two or more single bonds. … Conjugated dienes have conjugated double bonds separated by one single bond. Cumulated dienes (cumulenes) have the double bonds sharing a common atom as in a group of compounds called allenes.



