Why is osmosis low to high
In diffusion, particles move from an area of higher concentration to one of lower concentration until equilibrium is reached. In osmosis, a semipermeable membrane is present, so only the solvent molecules are free to move to equalize concentration.
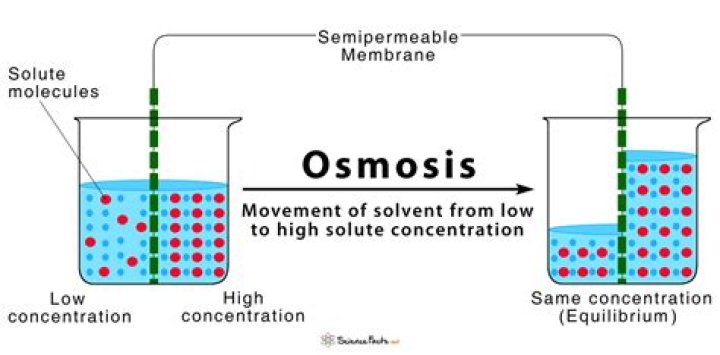
Does water move from high to low in osmosis?
1: Osmosis: In osmosis, water always moves from an area of higher water concentration to one of lower concentration. … In this example, the solute cannot diffuse through the membrane, but the water can. Water has a concentration gradient in this system.
What is a high concentration and low concentration?
Diffusion is the net movement of molecules from an area where they are at a higher concentration to areas where they are at a lower concentration. This is due to the random movement of the molecules. The difference in the concentration of a substance between two areas is called the concentration gradient .
What is the movement from low to high concentration?
DiffusionWhat is it?Diffusion is a spontaneous movement of particles from an area of high concentration to an area of low concentration. (ex. tea flavoring moving from an area of high to low concentration in hot water.)Why does water move from high to low water potential?
Water always moves from the system with a higher water potential to the system with a lower water potential. Solute potential (Ψs) decreases with increasing solute concentration; a decrease in Ψs causes a decrease in the total water potential.
Where does osmosis take place?
Osmosis occurs in both the small and large intestines, with the majority of osmosis occurring in the large intestine. As your body processes food, it moves from the esophagus to the stomach and then to the small intestine. While there, your body absorbs important nutrients via osmosis.
Which movement occurs by osmosis?
Osmosis is a special type of diffusion, namely the diffusion of water across a semipermeable membrane. Water readily crosses a membrane down its potential gradient from high to low potential (Fig. 19.3) [4]. Osmotic pressure is the force required to prevent water movement across the semipermeable membrane.
What is specific to osmosis?
CC BY-NC-SA) Osmosis. Osmosis is a specific type of diffusion; it is the passage of water from a region of high water concentration through a semi-permeable membrane to a region of low water concentration.Does the rate of osmosis increase with concentration?
The concentration of a solute affects the rate of osmosis over time, in a way where, the higher the concentration of a solute, the faster the rate of osmosis. This happens because, in a semi-permeable membrane the water is the only through that can move through. … That results in the rate of osmosis being faster.
What's the opposite of osmosis?Active transport requires energy. This is because in active transport, a substance is moved from an area of low concentration to an area of high concentration, against the concentration gradient. This is the opposite direction to diffusion and osmosis.
Article first time published onHow do you remember osmosis?
An Australian woman, Oz for short, only mows (osmosis) her lawn in the rain (water). When she does so, the grass changes from being very high to being very low.
Is osmosis a form of diffusion?
Osmosis is a special type of diffusion, namely the diffusion of water across a semipermeable membrane.
Does water have a high water potential?
It can also be described as a measure of how freely water molecules can move in a particular environment or system. It is measured in kilopascals (kPa) and is represented by the Greek letter Psi (Ψ). Water potential is never positive but has a maximum value of zero, which is that of pure water at atmospheric pressure.
What is the difference between high and low water potential?
For example, if water is at the top of a ramp, it has a high water potential due to gravity (it would tend to move downhill). … If there is a low water potential, then this means that there are few forces driving the water to move from one place to another, and the water will tend to remain as is.
Is high water potential hypertonic?
The addition of solute decreases water potential. … A hypertonic solution has lower water potential. A hypotonic solution has higher water potential.
Why does osmosis occur faster at higher temperatures?
Temperature – The higher the temperature, the faster the water molecules move across the semi permeable membrane. Surface Area – The larger the surface area, the more space for the molecules to move easily across; the smaller the area, the more restricted the movements of the molecules and the slower the movement.
Where is the low concentration of perfume?
The perfume is at its highest concentration in the bottle and is at its lowest at the edges of the room. The perfume vapor will diffuse, or spread away, from the bottle, and gradually, more and more people will smell the perfume as it spreads.
Is diffusion always from high to low concentration?
Diffusion. Diffusion is a passive process of transport. A single substance tends to move from an area of high concentration to an area of low concentration until the concentration is equal across a space. You are familiar with diffusion of substances through the air.
What is osmosis 9th class?
Osmosis is the movement of water molecules or a solvent from a region of low water concentration towards a region of high water concentration of solute through a semi-permeable membrane. Osmosis is a vital process in biological systems, which occurs in liquids, supercritical liquids and gases.
Does osmosis increase mass?
When the chips were put in distilled water they gain mass because the chips gain water from the surrounding solution due to osmosis. The process of osmosis causes a net flow of water across the semi permeable membrane, from a solution with a high water concentration to one with a lower water concentration.
Does higher concentration mean higher osmotic pressure?
Osmotic pressure is affected by concentration and temperature. Concentration of solute and temperature each affect the amount of pressure created by the movement of water across a membrane. Higher concentrations and higher temperatures increase osmotic pressure.
Which statement about osmosis is correct?
Which of the following statements about osmosis is correct? Osmosis is the diffusion of water from a region of lower water concentration to a region of higher water concentration. Osmotic movement of water into a cell would likely occur if the cell accumulates water from its environment.
Is low to high concentration active or passive?
During active transport, substances move against the concentration gradient, from an area of low concentration to an area of high concentration. This process is “active” because it requires the use of energy (usually in the form of ATP). It is the opposite of passive transport.
What is a high concentration?
A high concentration of a substance in a solution means that there’s a lot of it relative to the volume: the Great Salt Lake has very few fish because of the high concentration of salt. To say that you have good concentration skills means that you pay attention well. Definitions of concentration.
Does osmosis apply to gases?
Description. Osmosis is the movement of a solvent across a semipermeable membrane toward a higher concentration of solute. In biological systems, the solvent is typically water, but osmosis can occur in other liquids, supercritical liquids, and even gases.
How do you explain osmosis to a child?
Osmosis allows water to pass through cell membranes. The transfer of water from a dilute solution to a concentrated solution over a partly permeable membrane is defined as osmosis.
What is osmosis grade 8?
Osmosis is the diffusion of water through a semi-permeable membrane.[1] More. specifically, it is the movement of water across a semi-permeable membrane from an. area of high water potential (low solute concentration) to an area of low water. potential (high solute concentration).
What is osmosis in simple words?
osmosis, the spontaneous passage or diffusion of water or other solvents through a semipermeable membrane (one that blocks the passage of dissolved substances—i.e., solutes).
Is osmosis faster than diffusion?
Diffusion across a membrane depends on the size and electric charge of molecules. Smaller molecules diffuse faster than larger molecules. … Osmosis is the flow of water molecules and depends on the particle concentration — not the type of molecule on either side of the membrane.
What energy drives osmosis?
Osmosis is a selective diffusion process driven by the internal energy of the solvent molecules. It is convenient to express the available energy per unit volume in terms of “osmotic pressure”. It is customary to express this tendency toward solvent transport in pressure units relative to the pure solvent.
What causes osmosis?
Osmosis occurs according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes. … Osmosis occurs when there is a concentration gradient of a solute within a solution, but the membrane does not allow diffusion of the solute.



