Why is pure metal not used
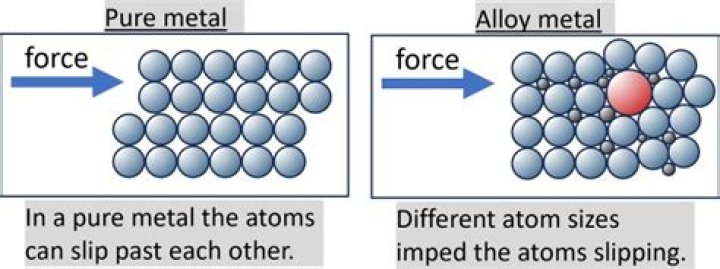
Most pure metals are either too soft, brittle, or chemically reactive for practical use. Combining different ratios of metals as alloys modifies the properties of pure metals to produce desirable characteristics.
What is the best metal to make bells out of?
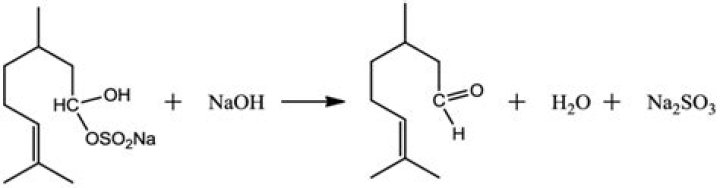
Bronze used for making bells and gongs is essentially an alloy of copper and tin. Copper containing about 22-24 wt% of tin is often known as bell metal because it has a pleasing sound quality when struck.
Why are bells not made up of non metals?
Metals have the property of producing a ringing sound when struck. Therefore, metals are sonorous and have the property of “Sonority”. Non-metals however, are not sonorous and hence are not used in bells. :D.
Why is pure copper not used for bells?
Give one property of copper. … Why isn’t pure copper used for bells instead of bronze? it is softer and can be bent; not the same sound. How much would you have to zoom in on a map of the United States to replicate the power of an electron microscope?How does purity of metal affect corrosion?
There are mainly two factors that affect corrosion: metallic and environmental. … Purity of metal: Pure metals are resistive to the corrosion. If there are impurities in a metal, a local galvanic cell is created with the metal as an anode and the impurity as a cathode. The result is corrosion of the metal.
Why is copper used for bells?
The metal used is a high-tin alloy of copper and tin with approximately a 4:1 ratio of copper to tin (78% copper, 22% tin). … It has been found that increasing the tin content increases the decay time of the bell strike, thus making the bell more sonorous.
Are alloys better than pure metals?
An alloy has better characteristics than a pure metal. … Adding other substances can make the pure metal stronger and/or corrosion resistant. This is the advantage of alloys as opposed to pure metals. One of the most common alloys is steel.
Who made crystal metal bells?
Iron Bell History. Bells were invented in 400 A.D. by Bishop Paulinus of Compagnia. They were not used in churches until some two hundred years later.How can you tell if bell is brass or bronze?
Properties of brass & bronze Brass can appear reddish, however, if the zinc has been removed from the surface metal by improper cleaning with harsh acids. Brass can also look red or pinkish if it is a “red” brass alloy. Bronze can look exactly like yellow brass or it may look almost like copper.
What metal are church bells made of?NARRATOR: The bell is made from bronze – an alloy consisting of 78 percent copper and 22 percent tin. The molten copper and tin is mixed by experts like Rudolf Perner, who know exactly how the metals react to heat.”
Article first time published onAre bells made of metal?
Bells made up of metals and not from wood because metals have the capability to produce sound when hit with a solid object, i.e, they are Sonorous.
Which metals dont corrode easily?
Gold and Platinum are the metals which do not corrode easily.
Do impurities affect corrosion?
In general the effect of impurities in the metal on the rate of corrosion was very slight. However, zinc-2yC-copper alloy sheet corroded less than pure zinc sheet.
Why impure metals corrode faster than pure metal?
Impure metals undergo the process of corrosion at faster pace than the pure metal. It is because, as the impure metals are composed of impurities, they are affected by condition called heterogeneity which leads to faster corrosion.
Is Aluminium an alloy or a pure metal?
Aluminium alloys (or aluminum alloys; see spelling differences) are alloys in which aluminium (Al) is the predominant metal. The typical alloying elements are copper, magnesium, manganese, silicon, tin and zinc.
Why are pure metals not used in dentistry?
In its pure form, most metals suffer from distinct disadvantages such as softness, brittleness or proneness to rust. This problem can be solved by combining individual metals, each giving a unique characteristic such as strength, corrosion resistance and other mechanical properties.
Is steel a pure metal?
As steel is an alloy, it is not a pure element and is, as a direct result, not actually a metal. Instead, it is actually a variant of a metal. Although steel is composed of iron – which is a metal – the non-metal carbon within its chemical make-up means that it is not a pure metal, so it cannot be classed as one.
Is Bell metal and bronze same?
composition and characteristics Bell metal, characterized by its sonorous quality when struck, is a bronze with a high tin content of 20–25 percent.
How can you tell how old a bell is?
- The edges of the bell will have become soft and irregular over time.
- The edges will show heavy wear and the rest of the bell will also look worn and seasoned.
- Antique bells will not have a casting or parting line. The new bells are typically cast in two pieces whereas antique farm bells were cast as one piece.
Which is more expensive brass or bronze?
Bronze is more expensive than brass. Zinc is cheaper than copper. The greater the zinc content the less the cost, and certain bronze alloys are four times more expensive than certain brass alloys.
Do magnets stick to brass?
In their natural states, metals such as brass, copper, gold, and silver will not attract magnets. This is because they are weak metals to start with.
How much is an old school bell worth?
Asking prices range from about $1,800 to $3,000, depending on the bells selected. They also stock bell parts for repairs and restoration. The American Bell Association has a forum for collectors of bells, including school house bells.
Is the CS Bell company still in business?
Bell Co. is no longer in Hillsboro – or in the bell- making business for that matter – C.S. Bell Co. bells are still being produced to this very day. You may or may not be aware that a company called Prindle Station, located in Washougal, Wash., manufactures C.S.
Should you paint an antique farm bell?
New bells are painted in the traditional color, black; but you can choose any paint color. Another method would be to protect the clean metal surface with a coat of WD-40. This would maintain the appearance of the metal and still protect it; however, a coat of WD-40 would need to be reapplied every year.
What are bells made of metal and not from wood?
The bells are made out of metal and not wood because metals are sonorous, have elastic-like properties, and can sustain vibrations for longer durations than wood.
Does Bell metal contain copper?
Bell metal is a hard alloy used for making bells and related instruments, such as cymbals. It is a form of bronze with a higher tin content, usually in approximately a 4:1 ratio of copper to tin (typically, 78% copper, 22% tin by mass).
Does copper and zinc make brass?
Brass is mainly an alloy that consists of copper with zinc added. Brasses can have varying amounts of zinc or other elements added. These varying mixtures produce a wide range of properties and variation in color.
Why do metals get corroded?
General corrosion occurs when most or all of the atoms on the same metal surface are oxidized, damaging the entire surface. Most metals are easily oxidized: they tend to lose electrons to oxygen (and other substances) in the air or in water. As oxygen is reduced (gains electrons), it forms an oxide with the metal.
Is gold corroded by air water and acid?
(d) gold is not corroded by air, water and acid. Explanation: Metals such as gold and platinum do not corrode visible because they are unaffected by oxygen and most acids.
Does aluminium get rust?
Rust is a type of corrosion (the wearing-away of metal), and to put it simply, aluminium does not rust, but it does corrode. Although these terms are often used interchangeably, they are fundamentally different. As with any metal, when it comes into contact with oxygen, an oxide layer will form on aluminium.
How does pH affect corrosion?
The corrosion rate tends to decrease with respect to pH; this means that corrosion rate is proportional to the solution’s acid concentration. The higher the concentration of nitric acid applied in the corrosion solution, the higher the corrosion rate is obtained.