Will an iron atom form a positive or negative ion
The yellow section, labeled “Transition Elements” are elements that tend to lose electrons from shells other than the outermost shell and form positive ions. For example, iron (Fe) forms two different positive ions, Fe+2 (ferrous iron) and Fe+3 (ferric iron).
Is iron positive or negative ion?
Halogens always form anions, alkali metals and alkaline earth metals always form cations. Most other metals form cations (e.g. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. oxygen, carbon, sulfur).
Is iron a negative charge?
For example, iron(II) has a 2+ charge; iron(III) a 3+ charge.
Will the atom form a positive or negative ion?
Neutral atoms can be turned into positively charged ions by removing one or more electrons. … Atoms that gain extra electrons become negatively charged. A neutral chlorine atom, for example, contains 17 protons and 17 electrons. By adding one more electron we get a negatively charged Cl- ion with a net charge of -1.How does an atom of iron form a positively charged iron?
Ions form when atoms gain or lose electrons. Since electrons are negatively charged, an atom that loses one or more electrons will become positively charged; an atom that gains one or more electrons becomes negatively charged. … The metals form positively-charged ions and the non-metals form negatively-charged ions.
Which atom will form a negatively charged ion?
Negatively-charged ions are anions, formed from nonmetallic elements like oxygen and sulfur.
How does an atom become a negative ion?
The atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called an anion). Opposite charges attract one another while similar charges repel one another.
Does iron have a set charge?
Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron(II) has a 2+ charge; iron(III) a 3+ charge.Which atom will form single positively charged ion?
EC : 2(K),1(L). It can lose one electron to gain inert gas configuration . hence it form Singly positively charged ion.
How do you know if an atom is positive or negative?If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons,it is a positive ion.
Article first time published onAre metals positively charged?
Metals usually form positive ions because metals are very “electropositive”. That means that the outer electrons of each atom of a metal are very loosely bound to the atom’s nucleus.
How does an iron atom change into an iron ion?
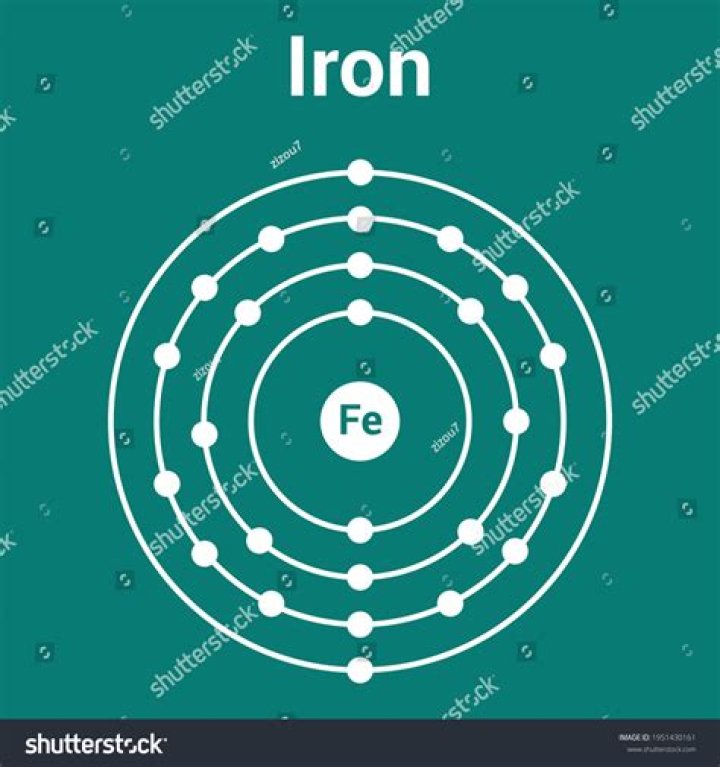
A neutral iron atom has 26 protons and 26 electrons. In order to form a 3+ ion, it must lose three electrons. The Fe atom loses two 4s electrons and one 3d electron.
Is iron atom or molecule?
Iron is a chemical element in the periodic table that has the symbol Fe and atomic number 26. Iron is group 8 and period 4 metal. A typical iron atom has 56 times the mass of a typical hydrogen atom.
How does an atom form an ion?
Ions are formed when the number of protons in an atom does not equal the number of electrons. If more protons are present, the ion is positive and is known as a cation; if more electrons are present, the ion is negative and referred to as an anion.
How does an atom become a positive ion a negative ion quizlet?
A positive ion has lost its valence electrons and has more protons (the number of protons is the number of the lost electrons) then electrons. A negative ion is when it has more electrons then protons. The chemical symbol for the element will change when it is charged.
What makes an atom an ion?
An ion is an atom or molecule that has a different number of electrons than protons, so it has a charge.
What is an example of a negative ion?
Examples of Negative Ions Fluoride is sometimes added to community water supplies. Your dentist may also give you a fluoride treatment. The negative charge (fewer protons than electrons) for an anion is shown by a number and minus sign after the formula. If there’s just a minus sign, it means the charge is minus 1.
What is a positively charged ion?
A cation is an ion with positive charge, which means it has more protons (positively-charged particles) than electrons (negatively-charged particles). Cations are formed when an atom loses one or more electrons: the loss of the negatively-charged electron(s) results in an overall positive charge.
How many neutrons are present in F '?
NucleusAtomic number, ZNumber of neutrons =A−Z613C613−6=7816O816−8=81224Mg1224−12=122656Fe2656−26=30
What's the ionic charge of iron?
NumberElementCharge26iron2+, 3+27cobalt2+, 3+28nickel2+29copper1+, 2+
What charge does an iron ion have?
ElementStemChargeironfe rr-2+3+coppercupr-1+2+
What is the positively charged atom?
ion, any atom or group of atoms that bears one or more positive or negative electrical charges. Positively charged ions are called cations; negatively charged ions, anions.
Is an atom a negative charge?
ParticleChargeMass (amu)Neutrons01.00867
What part of the atom has a positive charge?
The nucleus has an overall positive charge as it contains the protons.
Do metals always form positive ions?
metal atoms lose electrons to form positively charged ions. non-metal atoms gain electrons to form negatively charged ions.
Why are metal ions positive?
Metal atoms lose electrons from their outer shell when they form ions: the ions are positive, because they have more protons than electrons. the ions formed have full outer shells.
Can metals be negatively charged?
Metal atoms lose the electron, or electrons, in their highest energy level and become positively charged ions. Non-metal atoms gain an electron, or electrons, to become negatively charged ions. Carbonyl anions are mentioned below. Formally, the metal is in a negative oxidation state.
Why can iron form fe3+ and fe2+?
The reason for the two different charges is the fact that Iron has two stable ions, just one is more stable then the other. So electronic configuration of fe2+ becomes 1s2 2s2 2p6 3s2 3p6 3d6. Now for fe3+ we need to remove one extra electron from the outermost d orbital. i.e., fe3+- 1s2 2s2 2p6 3s2 3p6 3d5.
What is the difference between an atom and an iron?
As nouns the difference between atom and iron is that atom is the smallest, indivisible constituent part or unit of something while iron is (uncountable) a common, inexpensive metal, often black in color, that rusts, is attracted by magnets, and is used in making steel.
Is iron an element compound or mixture?
Elements. An element is a pure substance that cannot be broken down into different types of substances. Examples of elements include carbon, oxygen, hydrogen, and iron.
What is an iron atom made up of?
Iron atoms have 26 electrons and 26 protons with 30 neutrons occuring in the most abundant isotope. It is the sixth most abundant element in the universe. In its pure form iron is a fairly soft, grayish metal.