Why is Krypton a non metal
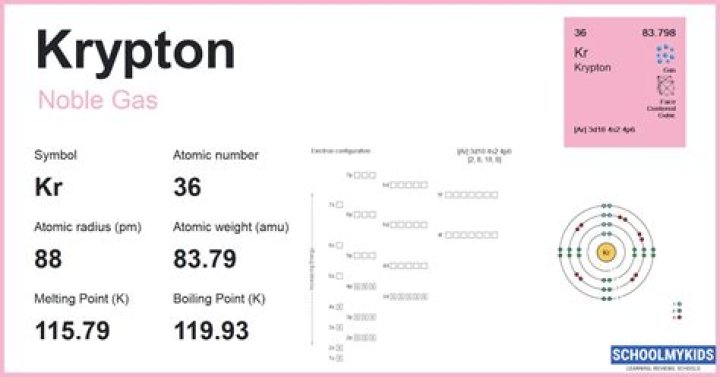
Classification:Krypton is a noble gas and a nonmetalState:gasMelting point:-157.3 oC, 115.9 KBoiling point:-153.2 oC, 119.4 KElectrons:36
Is Krypton a reactive nonmetal?
Uses and properties It does not react with anything except fluorine gas. Krypton is used commercially as a filling gas for energy-saving fluorescent lights. It is also used in some flash lamps used for high-speed photography. Unlike the lighter gases in its group, it is reactive enough to form some chemical compounds.
Is scandium a metal?
Uses and properties A silvery metal that tarnishes in air, burns easily and reacts with water. Scandium is mainly used for research purposes. It has, however, great potential because it has almost as low a density as aluminium and a much higher melting point.
What does Krypton belong to?
Krypton is chemical element number 36 on the periodic table of the elements. It belongs to the group of elements known as the noble gases. The other noble gases are helium, neon, argon, xenon, and radon. Under normal conditions, krypton is a colorless, tasteless, odorless gas.Is krypton an element compound or mixture?
It’s a noble gas with the atomic number of 36. It only consists of 1 type of atom and it cannot be broken down to produce other elements. Thus, krypton is neither a compound or a mixture. It’s a pure element.
Is krypton a metal or nonmetal or metalloid?
Krypton (Kr) exists as a colourless, odourless gas and is chemically inert. It has the atomic number 36 in the periodic table and belongs in Group 18, the Noble Gases. It is a non metal with the symbol Kr. Krypton like most Noble gases is odourless, colourless and inert.
Is krypton a compound?
krypton (Kr), chemical element, a rare gas of Group 18 (noble gases) of the periodic table, which forms relatively few chemical compounds. About three times heavier than air, krypton is colourless, odourless, tasteless, and monatomic.
Is krypton a conductor?
Krypton belongs to the family of noble gases. It has the atomic number 36. Therefore, krypton is an insulator they are poor conductors of heat and never conduct electricity. Krypton is classified as gas and non-metal.What type of solid is krypton?
Solid krypton is white and has a face-centered cubic crystal structure, which is a common property of all noble gases (except helium, which has a hexagonal close-packed crystal structure).
Is krypton a covalent network?No. Krypton does not form bonds with itself (or much of anything else). It has only Van Der Waals forces to form a liquid or solid.
Article first time published onWhich element is a noble gas?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
How is krypton in nature?
Although traces of krypton are found in various minerals, the most important source of krypton is Earth’s atmosphere. Air is also the most important source for the other noble gases, with the exception of helium (obtained from natural gas) and radon (obtained as a byproduct of the decay of radioactive elements).
Is yttrium a metal?
A soft, silvery metal. Yttrium is often used as an additive in alloys. It increases the strength of aluminium and magnesium alloys. It is also used in the making of microwave filters for radar and has been used as a catalyst in ethene polymerisation.
Is scandium an element?
Scandium is a chemical element with the symbol Sc and atomic number 21. A silvery-white metallic d-block element, it has historically been classified as a rare-earth element, together with yttrium and the lanthanides.
Is polonium a metal?
Polonium has a position in the periodic table that could make it a metal, a metalloid or a nonmetal. It is classed as a metal as its electrical conductivity decreases as its temperature rises.
Is a sausage a compound?
Column AColumn BColumn C1. Summer SausageHMChunky, Brown2. SteamCGas, Hot3. Salt WaterSLiquid, Clear4. Pencil lead (Pb)EGrey, Solid
Is Diamond an element compound or mixture?
It’s a heterogeneous mixture. Diamond is made of just one element: carbon. Each carbon atom in diamond is connected to four other carbon atoms, in a crystal that extends on and on. There are other forms of pure carbon where the atoms are bonded differently, notably charcoal and graphite.
What is heterogeneous mixture?
A heterogeneous mixture is a mixture in which the composition is not uniform throughout the mixture. … A heterogeneous mixture consists of two or more phases. When oil and water are combined, they do not mix evenly, but instead form two separate layers. Each of the layers is called a phase.
Is ammonia an element compound or mixture?
Ammonia is a compound of nitrogen and hydrogen with the formula NH3.
What are the common compounds of krypton?
- Krypton Tetrachloride. KrCl4. 225.61.
- Krypton Difluoride. KrF2. 121.7948.
- Krypton Hexabromine. KrBr6. 563.222.
- Krypton Dichromate. Kr2Cr2O7. 383.584.
- Krypton Chromate. KrCrO4. 199.7917.
- Krypton Tetrafluoride. KrF4. 159.7916128.
- Krypton Dioxide. KrO2. 115.7968.
Is antimony a metal or nonmetal?
Antimony is a semi-metal. In its metallic form it is silvery, hard and brittle. Antimony is used in the electronics industry to make some semiconductor devices, such as infrared detectors and diodes. It is alloyed with lead or other metals to improve their hardness and strength.
Is antimony a metalloid?
Elemental antimony is a brittle, silver-white, shiny metalloid.
What is the neutron of Krypton?
Krypton has 48 neutrons. The atomic number for krypton is 36. This number also tells you how many protons are in one atom of krypton.
Is krypton malleable or not malleable?
If you cool krypton down to the point that it solidifies (which is actually relatively easy: its freezing point is higher than the boiling point of liquid nitrogen), then you can convincingly argue that krypton is non-ductile, being quite brittle instead.
Is KR ionic or covalent?
Additionally, all of the halogen elements, including fluorine, chlorine, bromine, iodine and astatine, are all covalent nonmetal elements. The extremely stable noble gasses, including helium, neon, argon, krypton, xenon and radon, are all also nonmetal covalent elements.
What are molecular solids examples?
Molecular solids have low melting (Tm) and boiling (Tb) points compared to metal (iron), ionic (sodium chloride), and covalent solids (diamond). Examples of molecular solids with low melting and boiling temperatures include argon, water, naphthalene, nicotine, and caffeine (see table below).
What is the conductivity of krypton?
OverviewNameKryptonRefractive Index1.000427Speed of Sound1120 m/sThermal Conductivity0.00943 W/(m K)
Is krypton a insulator?
Argon and krypton are odorless, colorless, non-toxic inert gases that can be used instead of air between panes of glass to increase insulation and energy efficiency. Argon is the cheaper, more readily available gas, but Krypton is a better insulator.
Is krypton invisible?
Krypton, as has been established, is a colorless, odorless, tasteless gas. … The discoverers of krypton (Ramsay and Travers) also discovered helium, argon, xenon and neon.
What type of solid is XE?
atomic number54atomic weight131.29melting point−111.9 °C (−169.4 °F)boiling point−108.0 °C (−162.4 °F)
Is KR an ionic solid?
Ionic solids form when electrostatic attraction causes anions and cations to form a crystal lattice. … Ionic crystals are extremely stable because considerable energy is required to break ionic bonds.